Mechanistic study of the therapeutic effect of Portulaca oleracea L on colorectal inflammatory cancer transformation in mice by inhibiting Caspase 12 protein based on proteomics
-
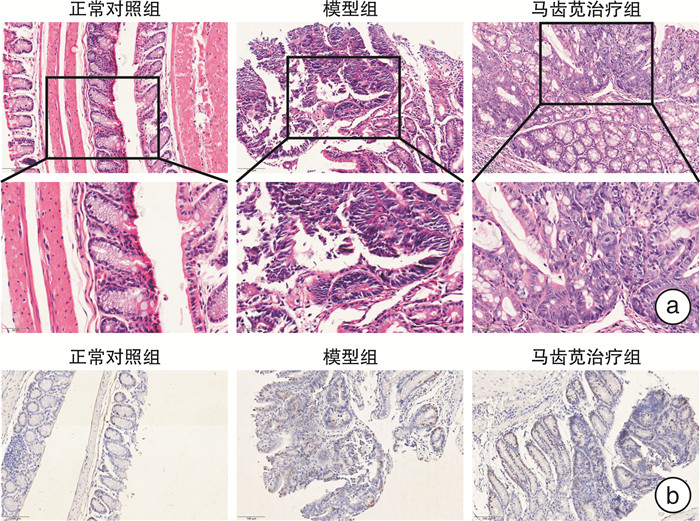
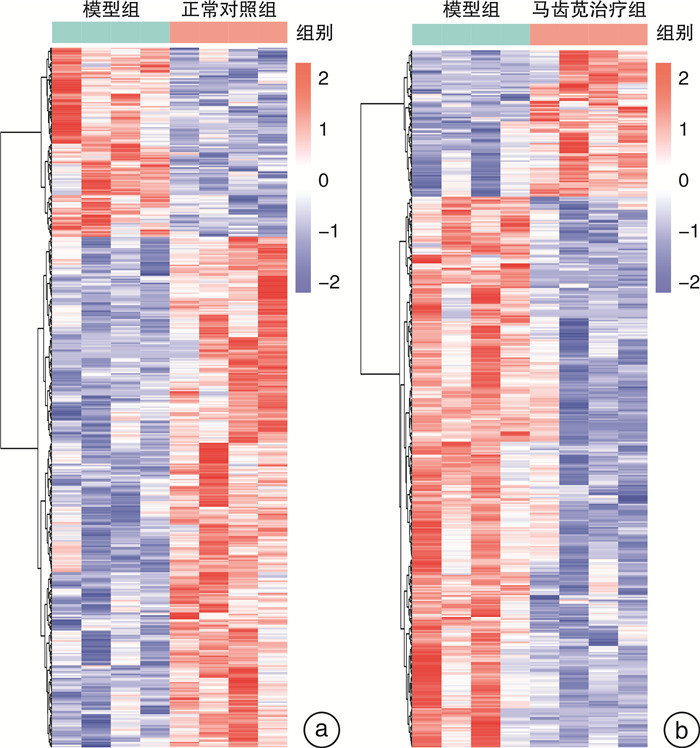
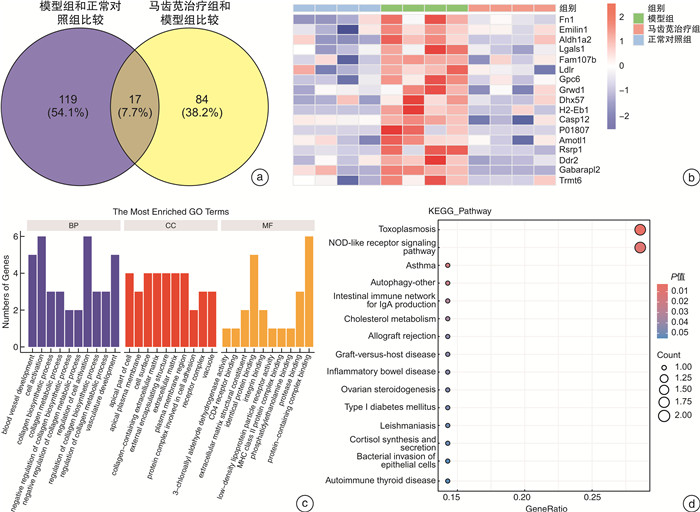
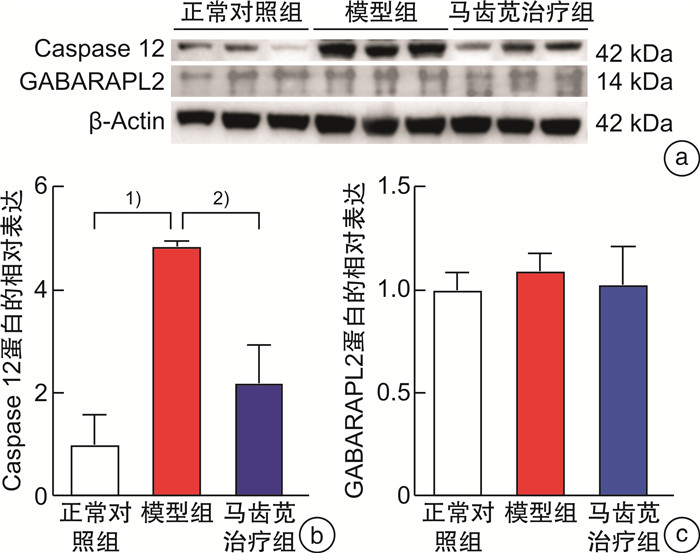
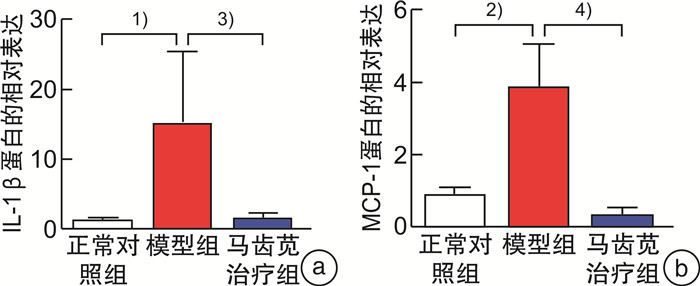
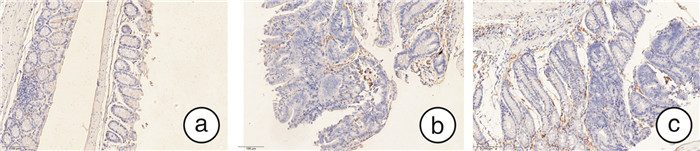
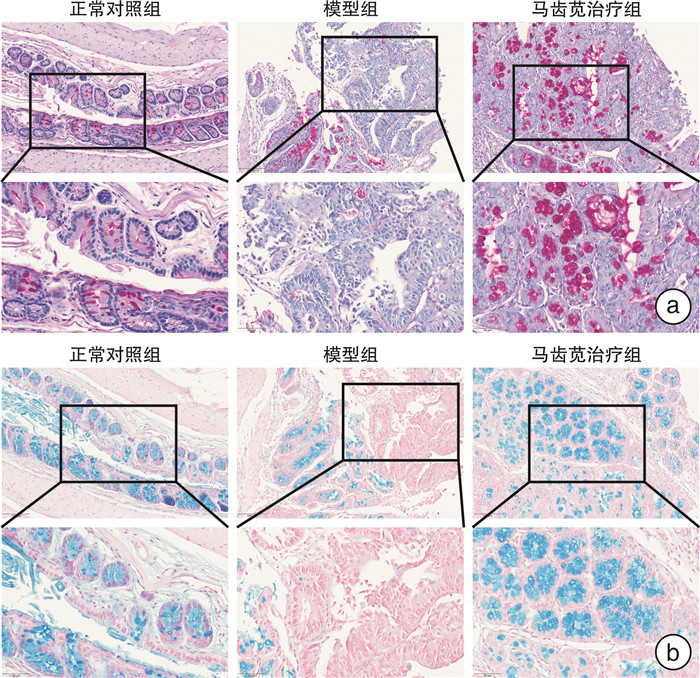
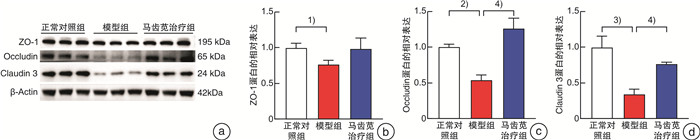
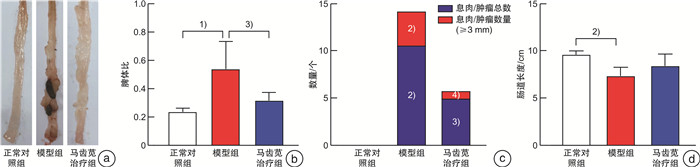
摘要: 目的 探讨马齿苋通过Caspase 12介导的NOD信号通路对结直肠炎癌转化小鼠的保护作用机制。方法 将24只6~8周龄的C57小鼠,随机分为正常对照组、模型组、马齿苋治疗组。模型组和马齿苋治疗组采用氧化偶氮甲烷(AOM)联合葡聚糖硫酸钠(DSS)诱导结直肠炎相关肿瘤发生小鼠模型,马齿苋治疗组每日按照人鼠等效剂量灌胃;通过各组小鼠的脾体比、结直肠长度、肠道息肉/肿瘤总数及肠道组织的苏木精-伊红染色,分析马齿苋抑制小鼠结直肠炎癌转化的药效;通过蛋白组分析、Western blot检测,验证马齿苋对NOD信号通路关键蛋白Caspase 12的调控;分别通过qPCR法和免疫荧光法检测小鼠组织中Caspase 12调控的细胞因子IL-1β、趋化因子和巨噬细胞标志物F4/80的表达;最后采用Alican blue和PAS病理染色检测肠道黏蛋白的水平,使用Western blot检测肠道紧密连接蛋白的水平,以评价马齿苋对肠道屏障的保护作用。结果 马齿苋明显抑制小鼠结直肠炎癌转化进展,减少肠道息肉/肿瘤数量和肠道Ki67水平,马齿苋可显著改善结直肠炎癌转化小鼠NOD样受体信号通路功能,抑制关键蛋白Caspase 12的水平,减少下游细胞因子IL-1β和趋化因子MCP-1含量,减少结直肠炎癌转化小鼠中肠道组织巨噬细胞的表达,改善结直肠炎癌转化小鼠肠道屏障功能。结论 马齿苋能够抑制小鼠结直肠炎癌转化进展,抑制炎症反应,改善肠道屏障,其作用可能与调控Caspase 12介导的NOD样受体信号通路有关。
-
关键词:
- 结直肠炎癌转化 /
- 马齿苋 /
- Caspase 12 /
- 肠道屏障
Abstract: Objective To investigate the mechanism of the protective effect of Portulaca oleracea L on colorectal inflammatory cancer transformation model mice through the NOD signaling pathway mediated by Caspase 12.Methods Twenty-four C57 mice aged 6-8 W were randomly divided into normal control, model control, and portulaca oleracea L treatment groups. In the model control group and portulaca oleracea L treatment group, Azoxymethane (AOM) combined with Dextran sodium sulfate (DSS) was used to induce colitis-associated tumorigenesis mice model, and in the portulaca oleracea L treatment group, gavage the drug every day according to the equivalent dose of human mice. The efficacy of Portulaca oleracea L in inhibiting colorectal cancer transformation mice was analyzed by ratio of spleen/body, colorectal length, total number of polyps/tumors, and HE staining of intestinal tissues in each group. Proteomic analysis and Western blot were used to clarify the therapeutic effect of the NOD signaling pathway, especially the key protein Caspase 12. The expression of cytokine IL-1β and macrophage marker F4/80 in mice tissues were detected by qPCR and immunofluorescence, respectively. Finally, the level of intestinal mucin was detected by Alican blue and PAS pathological staining, and the level of intestinal tight junction protein was detected by Western blot, to evaluate the protective effect of Portulaca oleracea L on the intestinal barrier.Results Portulaca oleracea L significantly inhibited the progression of colorectal inflammatory cancer transformation in mice, and reduced the number of intestinal polyps/tumors and intestinal Ki67 levels. Portulaca oleracea L significantly improved the function of the NOD-like receptor signaling pathway in colorectal inflammatory cancer transformation mice, inhibited the level of the key protein Caspase 12, reduced the levels of the downstream cytokine IL-1β and chemokine MCP-1, and inhibited the expression of macrophages in the intestinal tissues. Portulaca oleracea L improved the intestinal barrier function in colorectal inflammation cancer-transformed mice.Conclusion Portulaca oleracea L significantly inhibited the progression of colorectal inflammatory cancer transformation in mice models, suppressed inflammatory responses, and improved the intestinal barrier, and its effects may be related to the modulation of the Caspase 12-mediated NOD-like receptor signaling pathway. -

-
[1] Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249. doi: 10.3322/caac.21660
[2] 中华医学会肿瘤学分会, 国家卫生健康委员会医政司. 中国结直肠癌诊疗规范(2023版)[J]. 协和医学杂志, 2023, 14(4): 706-733. https://www.cnki.com.cn/Article/CJFDTOTAL-XHYX202304006.htm
[3] Ciemins EL, Mohl JT, Moreno CA, et al. Development of a Follow-Up Measure to Ensure Complete Screening for Colorectal Cancer[J]. JAMA Netw Open, 2024, 7(3): e242693. doi: 10.1001/jamanetworkopen.2024.2693
[4] Hisano K, Mizuuchi Y, Ohuchida K, et al. Microenvironmental changes in familial adenomatous polyposis during colorectal cancer carcinogenesis[J]. Cancer Lett, 2024, 589: 216822. doi: 10.1016/j.canlet.2024.216822
[5] Schmitt M, Greten FR. The inflammatory pathogenesis of colorectal cancer[J]. Nat Rev Immunol, 2021, 21(10): 653-667. doi: 10.1038/s41577-021-00534-x
[6] Ahmad S, Ashktorab H, Brim H, et al. Inflammation, microbiome and colorectal cancer disparity in African-Americans: Are there bugs in the genetics?[J]. World J Gastroenterol, 2022, 28(25): 2782-2801. doi: 10.3748/wjg.v28.i25.2782
[7] 魏小曼, 耿雪尘, 李柳, 等. 基于癌毒病机理论的结直肠癌全程防治体系构建[J]. 中华中医药杂志, 2023, 38(2): 572-575. https://www.cnki.com.cn/Article/CJFDTOTAL-BXYY202302095.htm
[8] 杨越, 郭亚芳, 余明华, 等. 430例晚期结直肠癌中医证型分布规律的回顾性研究[J]. 世界中西医结合杂志, 2023, 18(10): 2048-2054. https://www.cnki.com.cn/Article/CJFDTOTAL-SJZX202310025.htm
[9] 刘稼玺, 孙凌云, 彭蓉晏, 等. 基于中医证型探讨早中期结直肠癌患者根治术后心理负担与肠道菌群分布的关系[J]. 北京中医药, 2023, 42(10): 1076-1082. https://www.cnki.com.cn/Article/CJFDTOTAL-BJZO202310005.htm
[10] 付力, 王菲叶, 许云, 等. 结直肠癌中医证候分布及演变规律研究[J]. 中医肿瘤学杂志, 2024, 6(1): 63-69. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYZL202401010.htm
[11] 曾夏诗, 樊冬梅. 肠息肉中医证型分布及与大便性状改变相关性研究和危险因素分析[J]. 广州中医药大学学报, 2023, 40(11): 2708-2715. https://www.cnki.com.cn/Article/CJFDTOTAL-REST202311004.htm
[12] 唐舒高, 王佳佳, 田紫若, 等. 单味中药有效成分治疗溃疡性结肠炎研究进展[J]. 中国中医药图书情报杂志, 2024, 48(1): 234-238. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYTQ202401042.htm
[13] 赵蕊, 蔡亚平, 陈志宝, 等. 马齿苋多糖对老龄荷瘤小鼠抗宫颈癌的作用[J]. 中国老年学杂志, 2013, 33(18): 4480-4482. https://www.cnki.com.cn/Article/CJFDTOTAL-ZLXZ201318048.htm
[14] 张秀玲, 黄克强, 张锡流, 等. 马齿苋抗恶性肿瘤的研究进展[J]. 广西医学, 2021, 43(24): 2995-2999. https://www.cnki.com.cn/Article/CJFDTOTAL-GYYX202124021.htm
[15] 王倩, 范文涛. 马齿苋对白介素-6及溃疡性结肠炎癌变的影响[J]. 河南中医, 2013, 33(8): 1327-1328. https://www.cnki.com.cn/Article/CJFDTOTAL-HNZY201308079.htm
[16] 梁啸, 李亚楠, 王志娜, 等. 马齿苋物质基础及其药理活性研究进展[J]. 辽宁大学学报(自然科学版), 2023, 50(4): 373-384. https://www.cnki.com.cn/Article/CJFDTOTAL-LNDZ202304007.htm
[17] 王倩, 范文涛. 马齿苋多糖对溃疡性结肠炎相关性结肠癌IL-6/STAT3信号通路的影响[J]. 世界中医药, 2013, 8(10): 1256-1258. https://www.cnki.com.cn/Article/CJFDTOTAL-SJZA201310053.htm
[18] Wang M, Chen S, He X, et al. Targeting inflammation as cancer therapy[J]. J Hematol Oncol, 2024, 17(1): 13. doi: 10.1186/s13045-024-01528-7
[19] Shah SC, Itzkowitz SH. Colorectal Cancer in Inflammatory Bowel Disease: Mechanisms and Management[J]. Gastroenterology, 2022, 162(3): 715-730.e3. doi: 10.1053/j.gastro.2021.10.035
[20] Liu L, Dong W, Wang S, et al. Deoxycholic acid disrupts the intestinal mucosal barrier and promotes intestinal tumorigenesis[J]. Food Funct, 2018, 9(11): 5588-5597. doi: 10.1039/C8FO01143E
[21] Leoncini G, Cari L, Ronchetti S, et al. Mucin Expression Profiles in Ulcerative Colitis: New Insights on the Histological Mucosal Healing[J]. Int J Mol Sci, 2024, 25(3): 1858. doi: 10.3390/ijms25031858
[22] Quansah E, Gardey E, Ramoji A, et al. Intestinal epithelial barrier integrity investigated by label-free techniques in ulcerative colitis patients[J]. Sci Rep, 2023, 13(1): 2681. doi: 10.1038/s41598-023-29649-y
[23] Coll RC, O'Neill LA. New insights into the regulation of signalling by toll-like receptors and nod-like receptors[J]. J Innate Immun, 2010, 2(5): 406-421. doi: 10.1159/000315469
[24] 李园, 韩言言, 钟林翰, 等. 牛病毒性腹泻病毒Erns蛋白激活NOD样受体热蛋白结构域相关蛋白3炎症小体诱发细胞焦亡的研究[J]. 微生物学报, 2023, 63(11): 4271-4282. https://www.cnki.com.cn/Article/CJFDTOTAL-WSXB202311015.htm
[25] 方懿, 张亚磊, 符清瑶, 等. 豆蔻明通过激活芳香烃受体/核因子红系2相关因子2/NOD样受体热蛋白结构域相关蛋白3信号通路减轻脂多糖诱导的猪小肠上皮细胞的氧化应激和炎症反应[J]. 动物营养学报, 2024, 36(2): 1193-1203. https://www.cnki.com.cn/Article/CJFDTOTAL-DWYX202402048.htm
[26] 邓妃富, 魏劲松. 骨免疫学背景下的NOD样受体及炎症小体在关节炎中的研究现状[J]. 医学信息, 2024, 37(2): 187-192. https://www.cnki.com.cn/Article/CJFDTOTAL-YXXX202402040.htm
[27] LeBlanc PM, Yeretssian G, Rutherford N, et al. Caspase-12 modulates NOD signaling and regulates antimicrobial peptide production and mucosal immunity[J]. Cell Host Microbe, 2008, 3(3): 146-157. doi: 10.1016/j.chom.2008.02.004
[28] Scott AM, Saleh M. The inflammatory caspases: guardians against infections and sepsis[J]. Cell Death Differ, 2007, 14(1): 23-31. doi: 10.1038/sj.cdd.4402026
[29] 王双双, 郑佳露, 陈锦芳, 等. 藤龙补中汤对大肠癌LoVo细胞失巢凋亡的作用及机制[J]. 中国中西医结合消化杂志, 2023, 31(9): 686-690. https://zxyxh.whuhzzs.com/article/doi/10.3969/j.issn.1671-038X.2023.09.06
[30] 许广慈, 杨如意. 大肠癌患者癌因性疲乏的中西医治疗进展[J]. 中国中西医结合消化杂志, 2022, 30(9): 673-678. https://zxyxh.whuhzzs.com/article/doi/10.3969/j.issn.1671-038X.2022.09.12
-




 下载:
下载: