Effects of Qidi Tongbian Prescription on intestinal flora in mice with slow transit constipation based on pseudo-sterile experiment
-
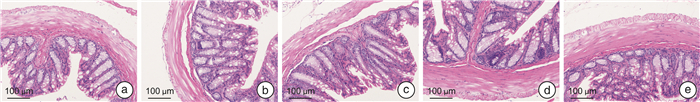
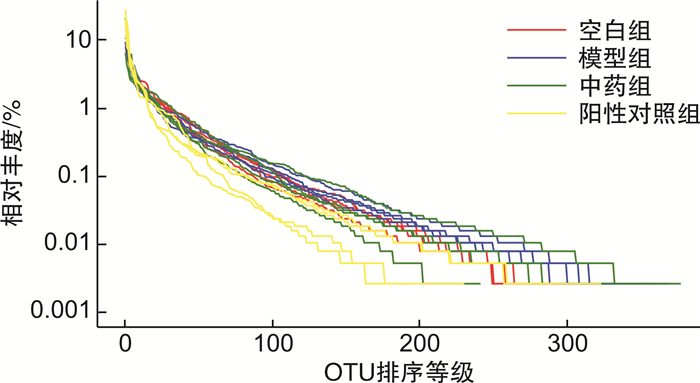
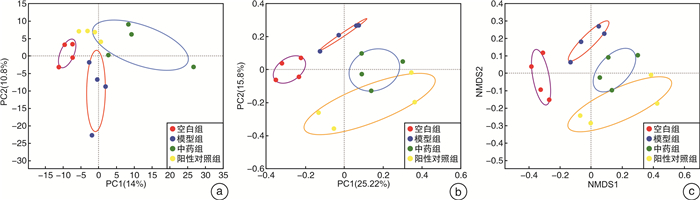
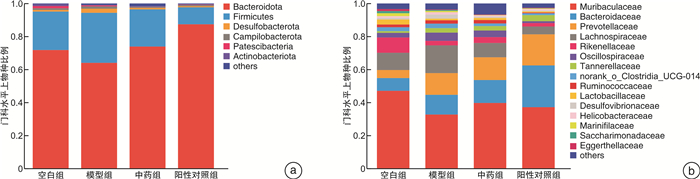
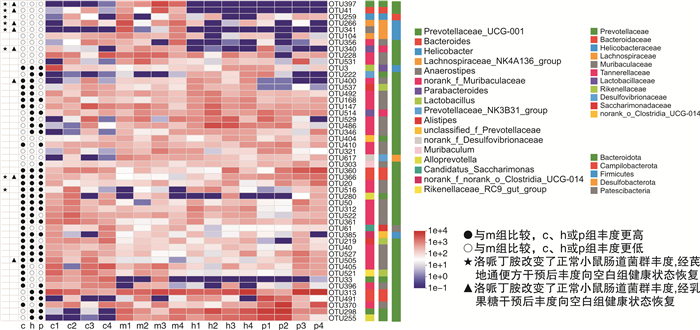
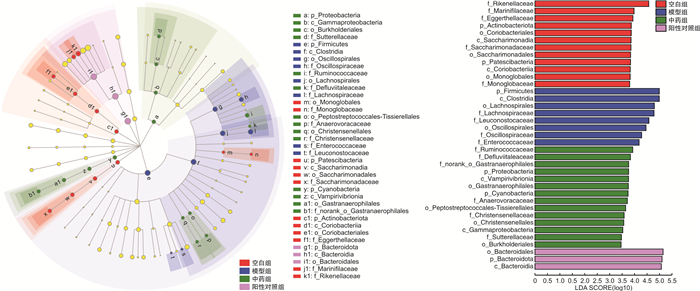
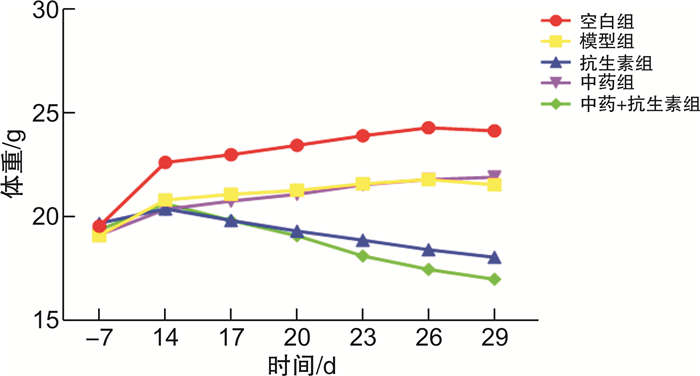
摘要: 目的 探索芪地通便方是否通过肠道菌群起到治疗慢传输型便秘(slow transit constipation,STC)的作用,并研究芪地通便方对STC小鼠肠道菌群的影响。方法 实验1将25只BALB/c小鼠分为5组,除空白组外盐酸洛哌丁胺(10 mg/kg)灌胃14 d造模,第15~28天,抗生素组予抗生素混合液自由饮,中药组予芪地通便方(18.906 g/kg)灌胃,中药+抗生素组予芪地通便方灌胃+抗生素混合液自由饮用。通过小鼠一般情况、体重、粪便含水率与数量、小肠推进率、结肠组织学和粪便DNA含量情况评价芪地通便方的疗效以及是否通过肠道菌群发挥作用。实验2将20只BALB/c小鼠分为4组,除空白组外盐酸洛哌丁胺(10 mg/kg)灌胃14 d造模,随后予芪地通便方(18.906 g/kg)和阳性药物乳果糖(3 mg/kg)灌胃干预14 d。通过16S rDNA基因测序探究小鼠肠道菌群的变化。结果 实验1发现抗生素组粪便DNA含量明显降低(P<0.05),证实本研究采用的抗生素混合液方法可有效消耗肠道菌群,成功建立肠道伪无菌小鼠。与模型组比较,中药组粪便含水率与数量、小肠推进率显著提高(P<0.05),而中药+抗生素组在以上2个方面差异无统计学意义(P>0.05),中药组和中药+抗生素组的疗效形成鲜明对比,反向验证了芪地通便方依赖于肠道菌群的存在从而达到治疗STC的作用。实验2发现芪地通便方能够改善STC小鼠的肠道菌群多样性及结构,尤其是能降低厚壁菌门,提高拟杆菌门丰度,降低厚壁菌门/拟杆菌门的比例。结论 芪地通便方治疗STC的疗效依赖于肠道菌群的存在,且该方可以调节STC小鼠肠道菌群。Abstract: Objective To explore whether Qidi Tongbian Prescription plays a role in the treatment of slow transit constipation(STC) through the presence of intestinal flora and to study the effect of Qidi Tongbian Prescription on intestinal flora of STC mice.Methods In experiment 1, 25 BALB/c mice were divided into 5 groups. Except the blank group, loperamide hydrochloride(10 mg/kg) was given intragastric administration for 14 days to establish the model. The antibiotic group was given a free drink of antibiotic mixture, the Chinese medicine group was given a Qidi Tongbian Prescription(18.906 g/kg) by gavage, and the Chinese medicine+antibiotics group was given a Qidi Tongbian Prescription by gavage + free drink of antibiotic mixture. The general condition, body weight, fecal moisture content and quantity, small intestinal propulsion rate, colon histology, and fecal DNA content of mice were used to evaluate the efficacy of Qidi Tongbian Prescription and whether it depended on the existence of intestinal flora. In experiment 2, 20 BALB/c mice were divided into 4 groups. Except for the blank group, loperamide hydrochloride(10 mg/kg) was administrated by gavage for 14 days to establish an STC mouse model, and then Qidi Tongbian Prescription(18.906 g/kg) and positive drug lactulose(3 mg/kg) were administrated by gavage for 14 days. The changes in intestinal flora in mice were investigated by 16S rDNA gene sequencing.Results In experiment 1, it was found that fecal DNA content in the antibiotic group was significantly decreased(P < 0.05), which confirmed that the antibiotic mixture method adopted in this study could effectively consume intestinal flora and successfully establish intestinal pseudo-aseptic mice.Compared with the model group, fecal moisture content, quantity, and small intestinal propulsion rate of the Qidi Tongbian Prescription group were significantly increased(P < 0.05), while there was no statistical difference in the above two aspects of Qidi Tongbian Prescription + antibiotic group(P > 0.05). The effect of the two groups was in stark contrast, which inversely verified that Qidi Tongbian Prescription was dependent on the presence of intestinal flora to treat STC. In experiment 2, it was found that Qidi Tongbian Prescription could improve the intestinal flora diversity and structure of STC mice, especially by reducing firmicutes, increasing the abundance of Bacteroides, and reducing the ratio of firmicutes to Bacteroides.Conclusion The effects of Qidi Tongbian Prescription on STC depended on the presence of intestinal flora, and it could regulate the intestinal flora of STC mice.
-

-
表 1 伪无菌小鼠第15~28天干预方法
组别 盐酸洛哌丁胺灌胃 无菌水灌胃 芪地通便方灌胃 无菌水自由饮用 抗生素混合液自由饮用 空白组 0.2 mL √ 模型组 0.1 mL 0.1 mL √ 抗生素组 0.1 mL 0.1 mL √ 中药组 0.1 mL 0.1 mL √ 中药+抗生素组 0.1 mL 0.1 mL √ 表 2 体重增加量、粪便含水率和数量、小肠推进率比较
X±S 组别 体重增加/g 粪便含水率/% 粪便数量/个 小肠推进率/% 空白组(n=5) 1.53±0.36 62.85±12.85 22.20±7.33 89.80±5.39 模型组(n=5) 0.74±0.471) 46.60±3.701) 11.60±4.671) 65.59±15.881) 抗生素组(n=5) -2.35±0.383) 51.45±11.33 13.60±4.93 54.86±8.58 中药组(n=5) 1.55±0.132) 61.75±7.842) 21.80±2.782) 87.97±12.942) 中药+抗生素组(n=5) -3.65±0.533)4) 53.94±5.93 16.40±5.13 67.55±10.654) 与空白组比较,1)P<0.05;与模型组比较,2)P<0.05,3)P<0.01;与中药组比较,4)P<0.01。 表 3 小鼠肠道菌群α多样性Ace指数
X±S 组别 Ace指数 空白组(n=4) 90.65±1.35 模型组(n=4) 102.17±1.251) 中药组(n=4) 111.66±5.732) 阳性对照组(n=4) 85.27±5.792) 与空白组比较,1)P<0.01;与模型组比较,2)P<0.05。 -
[1] 中华医学会消化病学分会胃肠动力学组, 功能性胃肠病协作组. 中国慢性便秘专家共识意见(2019, 广州)[J]. 中华消化杂志, 2019, 39(9): 577-598. doi: 10.3760/cma.j.issn.0254-1432.2019.09.001
[2] Bharucha AE, Lacy BE. Mechanisms, Evaluation, and Management of Chronic Constipation[J]. Gastroenterology, 2020, 158(5): 1232-1249.e3. doi: 10.1053/j.gastro.2019.12.034
[3] Barberio B, Judge C, Savarino EV, et al. Global prevalence of functional constipation according to the Rome criteria: a systematic review and meta-analysis[J]. Lancet Gastroenterol Hepatol, 2021, 6(8): 638-648. doi: 10.1016/S2468-1253(21)00111-4
[4] Chen Z, Peng Y, Shi Q, et al. Prevalence and Risk Factors of Functional Constipation According to the Rome Criteria in China: A Systematic Review and Meta-Analysis[J]. Front Med(Lausanne), 2022, 9: 815156.
[5] Tanner S, Chaudhry A, Goraya N, et al. Prevalence and Clinical Characteristics of Dyssynergic Defecation and Slow Transit Constipation in Patients with Chronic Constipation[J]. J Clin Med, 2021, 10(9): 2027. doi: 10.3390/jcm10092027
[6] 王娅琼. 结肠传输试验对慢性功能性便秘诊断和分型的临床研究[D]. 蚌埠: 蚌埠医学院, 2019.
[7] 崔文文, 管忠安. 中医诊治慢性便秘现状与进展[J]. 现代中西医结合杂志, 2021, 30(36): 4094-4099. doi: 10.3969/j.issn.1008-8849.2021.36.022
[8] Li H, Zhang P, Xue Y. A comparison of the safety and efficacy of polyethylene glycol 4000 and lactulose for the treatment of constipation in pregnant women: a randomized controlled clinical study[J]. Ann Palliat Med, 2020, 9(6): 3785-3792. doi: 10.21037/apm-20-1674
[9] Southwell BR. Treatment of childhood constipation: a synthesis of systematic reviews and meta-analyses[J]. Expert Rev Gastroenterol Hepatol, 2020, 14(3): 163-174. doi: 10.1080/17474124.2020.1733974
[10] Blaser MJ. Fecal Microbiota Transplantation for Dysbiosis-Predictable Risks[J]. N Engl J Med, 2019, 381(21): 2064-2066. doi: 10.1056/NEJMe1913807
[11] Sharma A, Rao S, Kearns K, et al. Review article: diagnosis, management and patient perspectives of the spectrum of constipation disorders[J]. Aliment Pharmacol Ther, 2021, 53(12): 1250-1267. doi: 10.1111/apt.16369
[12] Corsetti M, Brown S, Chiarioni G, et al. Chronic constipation in adults: Contemporary perspectives and clinical challenges. 2: Conservative, behavioural, medical and surgical treatment[J]. Neurogastroenterol Motil, 2021, 33(7): e14070. doi: 10.1111/nmo.14070
[13] Huang J, Li S, Wang Q, et al. Pediococcus pentosaceus B49 from human colostrum ameliorates constipation in mice[J]. Food Funct, 2020, 11(6): 5607-5620. doi: 10.1039/D0FO00208A
[14] 黄继汉, 黄晓晖, 陈志扬, 等. 药理试验中动物间和动物与人体间的等效剂量换算[J]. 中国临床药理学与治疗学, 2004, 9(9): 1069-1072. doi: 10.3969/j.issn.1009-2501.2004.09.026
[15] 杨志然, 李军祥, 陈润花, 等. 芪地通便方治疗功能性便秘患者的临床疗效分析[J]. 中国中西医结合消化杂志, 2022, 30(12): 820-825. doi: 10.3969/j.issn.1671-038X.2022.12.02
[16] Farmer AD, Holt CB, Downes TJ, et al. Pathophysiology, diagnosis, and management of opioid-induced constipation[J]. Lancet Gastroenterol Hepatol, 2018, 3(3): 203-212. doi: 10.1016/S2468-1253(18)30008-6
[17] Meng J, Abu YF, Zhang Y, et al. Opioid-induced microbial dysbiosis disrupts irinotecan(CPT-11) metabolism and increases gastrointestinal toxicity in a murine model[J]. Br J Pharmacol, 2023, 180(10): 1362-1378. doi: 10.1111/bph.16020
[18] Jalodia R, Abu YF, Oppenheimer MR, et al. Opioid Use, Gut Dysbiosis, Inflammation, and the Nervous System[J]. J Neuroimmune Pharmacol, 2022, 17(1-2): 76-93. doi: 10.1007/s11481-021-10046-z
[19] Huang J, Lin B, Zhang Y, et al. Bamboo shavings derived O-acetylated xylan alleviates loperamide-induced constipation in mice[J]. Carbohydr Polym, 2022, 276: 118761. doi: 10.1016/j.carbpol.2021.118761
[20] Bai J, Cai Y, Huang Z, et al. Shouhui Tongbian Capsule ameliorates constipation via gut microbiota-5-HT-intestinal motility axis[J]. Biomed Pharmacother, 2022, 154: 113627. doi: 10.1016/j.biopha.2022.113627
[21] 李建香, 王君君, 黄天马, 等. 无菌鼠模型制作方法与评价[J]. 实验动物科学, 2022, 39(3): 67-69, 73. https://www.cnki.com.cn/Article/CJFDTOTAL-SYDG202203013.htm
[22] Zhang X, Yang H, Zheng J, et al. Chitosan oligosaccharides attenuate loperamide-induced constipation through regulation of gut microbiota in mice[J]. Carbohydr Polym, 2021, 253: 117218. doi: 10.1016/j.carbpol.2020.117218
[23] Yu T, Ding Y, Qian D, et al. Characteristics of fecal microbiota in different constipation subtypes and association with colon physiology, lifestyle factors, and psychological status[J]. Therap Adv Gastroenterol, 2023, 16: 17562848231154101.
[24] Zhang X, Zheng J, Jiang N, et al. Modulation of gut microbiota and intestinal metabolites by lactulose improves loperamide-induced constipation in mice[J]. Eur J Pharm Sci, 2021, 158: 105676. doi: 10.1016/j.ejps.2020.105676
[25] Tian H, Chen Q, Yang B, et al. Analysis of Gut Microbiome and Metabolite Characteristics in Patients with Slow Transit Constipation[J]. Dig Dis Sci, 2021, 66(9): 3026-3035. doi: 10.1007/s10620-020-06500-2
[26] Wang L, Wang F, Zhang X, et al. Transdermal Administration of Volatile Oil from Citrus aurantium-Rhizoma Atractylodis Macrocephalae Alleviates Constipation in Rats by Altering Host Metabolome and Intestinal Microbiota Composition[J]. Oxid Med Cell Longev, 2022, 2022: 9965334.
[27] Müller M, Hermes G, Canfora EE, et al. Distal colonic transit is linked to gut microbiota diversity and microbial fermentation in humans with slow colonic transit[J]. Am J Physiol Gastrointest Liver Physiol, 2020, 318(2): G361-G369. doi: 10.1152/ajpgi.00283.2019
-




 下载:
下载: