Regulation mechanism of Qingchang Wenzhong Decoction on tissue-resident memory CD4+T cells in mice with ulcerative colitis
-
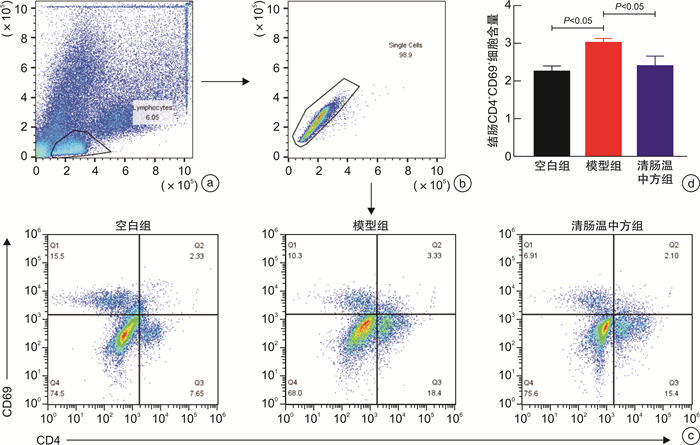
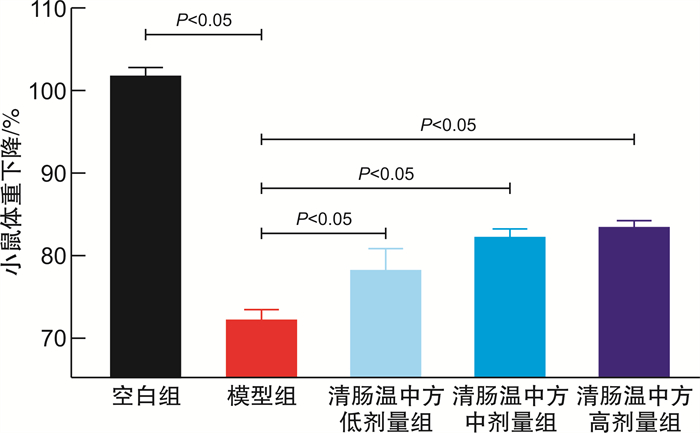
摘要: 目的 探究清肠温中方对溃疡性结肠炎(UC)小鼠组织驻留记忆CD4+T细胞(CD4+TRM细胞)的调控作用,为中医药治疗UC提供新的理论依据。方法 将SPF级健康雌性C57BL/6小鼠随机分为空白组、模型组、清肠温中方低剂量组、清肠温中方中剂量组、清肠温中方高剂量组。空白组小鼠全程自由进食饮水,模型组和清肠温中方低、中、高各剂量组均采用自由饮用2.5 %(w/v)DSS溶液7 d复制UC模型,同时模型组给予去离子水灌胃,清肠温中方各剂量组给予相应浓度的中药干预1周,期间每日观察小鼠一般情况,测量体重、检测便潜血、记录粪便性状,计算疾病活动指数(DAI)。干预结束后留取结肠组织,制备单细胞混悬液,流式细胞术检测CD4+TRM细胞的水平,应用RT-qPCR测定葡萄糖转运蛋白 Glut1、Glut2、Glut4 mRNA及糖酵解相关因子HK2、PKM2、G6PC、LDHA、LepR、HIF-1α mRNA的含量。结果 与空白组比较,UC小鼠呈现明显的肠道炎症,表现出不同程度的便血、腹泻、体重下降等,清肠温中方各剂量组小鼠体重较模型组明显升高(P < 0.05);模型组小鼠DAI较空白组升高明显(P < 0.05),清肠温中方各剂量组小鼠DAI下降明显(P < 0.05)。机制方面,模型组小鼠结肠固有层CD4+TRM细胞含量均明显高于空白组(P < 0.05),而经清肠温中方干预后,CD4+TRM细胞含量显著降低(P < 0.05);模型组小鼠的 Glut1、Glut2、Glut4、LepR、HIF-1α、HK2、PKM2、G6PC、LDHA mRNA水平较空白组升高(P < 0.05)或有升高趋势,清肠温中方组所检测的mRNA较模型组降低(P < 0.05)或有降低趋势。结论 清肠温中方可能通过抑制糖酵解代谢,从而调控CD4+TRM细胞免疫稳态,达到治疗UC的目的。
-
关键词:
- 溃疡性结肠炎 /
- 清肠温中方 /
- 组织驻留记忆CD4+T细胞 /
- 免疫调控
Abstract: Objective To explore the regulatory effect of Qingchang Wenzhong Decoction(QCWZD) on tissue-resident memory CD4+T cells(CD4+TRMcells) in mice with DSS-induced colitis.Methods Healthy female C57BL/6 mice were randomly divided into control group, model group, QCWZD low-dose group, QCWZD medium-dose group, QCWZD high-dose group. Mice in control group were fed and watered freely throughout the whole process. Model group and doses group of QCWZD were used to replicate the UC model by freely drinking 2.5 %(w/v) DSS solution for 7 days. At the same time, model group was given deionized water gavage, each dose group of QCWZD were given the corresponding concentration of QCWZD intervention for 1 week. During this period, the mice were observed daily for general condition, weight measurement, detection of fecal occult blood, recording of fecal properties, and calculation of disease activity index(DAI). After the intervention, colonic tissues were retained, single cell suspensions were prepared, and the levels of CD4+TRM cells were detected by flow cytometry. Application of RT-qPCR for the determination of glucose transporter proteins Glut1/2/4 mRNA and glycolysis-related HK2 mRNA, PKM2 mRNA, G6PC mRNA, LDHA mRNA, LepR mRNA, HIF-1α mRNA.Results Compared with control group, UC mice showed obvious intestinal inflammation, and exhibiting different degrees of blood in the stool, diarrhea and weight loss. The body weight of mice in each dose group of Chinese medicine were significantly higher than that of model group(P < 0.05). The DAI of mice in model group was significantly higher than that of control group(P < 0.05), and the DAI of mice in each dose group of the QCWZD decreased significantly(P < 0.05). Mechanistically, the content of CD4+TRM cells in the lamina propria of mice in the model group were significantly higher than those in the control group(P < 0.05). While the CD4+TRMcells content was significantly reduced(P < 0.05) after the intervention with the QCWZD. The levels of Glut1/2/4 mRNA, LepR mRNA, HIF-1αmRNA, HK2mRNA, PKM2mRNA, LDHA mRNA, G6PC mRNA were increased(P < 0.05) or tended to be decreased in model group mice compared with control group, and themRNA detected in QCWZD group were decreased(P < 0.05) or tended to be decreased compared with model group.Conclusion QCWZD may be used to treat UC by inhibiting glycolytic metabolism, thereby regulating CD4+TRMcells immune homeostasis. -

-
表 1 疾病活动指数的评分标准[12]
评分项目 0 1 2 3 4 体重下降指数 无下降 >1%~5% >5%~10% >10%~15% >15% 粪便性状 正常 / 松散 / 稀便 粪便潜血 无 / 阳性 / 肉眼血便 表 2 本实验所用引物序列
基因 上游(5′-3′) 下游(5′-3′) GAPDH CTTCCAGCCTTCCTTCCTTGG AATGCCTGGGTACATGGTGG LepR GGTCCAGGTGAGGAGCAAGA AAAGAAGCATTCGATCCAACACTA HIF-lα GGGTACAAGAAACCACCCAT GAGGCTGTGTCGACTGAGAA GlutI TCAACACGGCCTTCACTG CACGATGCTCAGATAGGACATC Glut2 TGTGCTGCTGGATAAATTCGCCTG AACCATGAACCAAGGGATTGGACC Glut4 GTAACTTCATTGTCGGCATGG AGCTGAGATCTGGTCAAACG HK2 TCGCCTGCTTATTCACGGAG CCATCCGGAGTTGACCTCAC G6PC GGTTCATCCTTGTGTCTGTG GCTGTAGTAGTCGGTGTCC PKM2 TGTCTGGAGAAACAGCCAAG CGAATAGCTGCAAGTGGTAGA LDHA CGTCTCCCTGAAGTCTCTTAAC TTCAGCTTGATCACCTCGTAG 表 3 清肠温中方对UC小鼠体重的影响
X±S 分组 例数 体重/g 空白组 5 18.82±0.47 模型组 5 13.56±0.841) 清肠温中方低剂量组 4 14.55±0.722) 清肠温中方中剂量组 5 15.74±0.582) 清肠温中方高剂量组 4 15.33±0.452) 与空白组比较,1)P < 0.05;与模型组比较,2)P < 0.05。 表 4 清肠温中方对UC小鼠DAI评分的影响
X±S 分组 例数 DAI 空白组 5 0 模型组 5 3.600±0.1631) 清肠温中方低剂量组 4 3.000±0.136 清肠温中方中剂量组 5 2.267±0.3862) 清肠温中方高剂量组 4 1.833±0.3192) 与空白组比较,1)P < 0.05;与模型组比较,2)P < 0.05。 表 5 清肠温中方对UC小鼠CD4+TRM细胞的调节作用
X±S 分组 例数 CD4+TRM细胞含量 空白组 5 2.276±0.118 模型组 5 3.063±0.0731) 清肠温中方组 4 2.423±0.2442) 与空白组比较,1)P < 0.01;与模型组比较,2)P < 0.05。 表 6 清肠温中方对UC小鼠结肠葡萄糖转运蛋白mRNA的影响
X±S 分组 例数 Glut1 mRNA Glut2 mRNA Glut4 mRNA 空白组 5 1.015±0.072 0.965±0.044 1.030±0.140 模型组 5 1.670±0.2041) 1.284±0.1231) 2.574±0.1051) 清肠温中方组 4 1.070±0.1422) 0.917±0.0782) 1.242±0.3912) 与空白组比较,1)P < 0.05;与模型组比较,2)P < 0.05。 表 7 清肠温中方对UC小鼠结肠糖酵解关键酶基因的调控作用
X±S 分组 例数 HK2 mRNA PKM2 mRNA G6PC mRNA LDHA mRNA 空白组 5 0.938±0.083 1.027±0.156 1.190±0.199 1.034±0.041 模型组 5 2.715±1.385 1.167±0.118 3.543±1.0171) 1.927±0.4721) 清肠温中方组 4 0.633±0.0642) 0.934±0.0242) 0.488±0.1772) 1.166±0.0472) 与空白组比较,1)P < 0.05;与模型组比较,2)P < 0.05。 表 8 清肠温中方对UC小鼠LepR/HIF-1α信号轴的调控作用
X±S 分组 例数 LepR mRNA HIF-1α mRNA 空白组 5 1.083±0.256 1.008±0.081 模型组 5 4.533±0.9561) 1.810±0.3821) 清肠温中方组 4 2.248±0.3432) 1.030±0.0582) 与空白组比较,1)P < 0.05;与模型组比较,2)P < 0.05。 -
[1] 李军祥, 陈誩. 溃疡性结肠炎中西医结合诊疗共识意见(2017年)[J]. 中国中西医结合消化杂志, 2018, 26(2): 105-111, 120. http://zxpw.cbpt.cnki.net/WKD2/WebPublication/paperDigest.aspx?paperID=b41ada9c-42e1-40f7-a3a2-410be59b996d
[2] Magro F, Gionchetti P, Eliakim R, et al. Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 1: Definitions, Diagnosis, Extra-intestinal Manifestations, Pregnancy, Cancer Surveillance, Surgery, and Ileo-anal Pouch Disorders[J]. J Crohns Colitis, 2017, 11(6): 649-670. doi: 10.1093/ecco-jcc/jjx008
[3] Raine T, Bonovas S, Burisch J, et al. ECCO Guidelines on Therapeutics in Ulcerative Colitis: Medical Treatment[J]. J Crohns Colitis, 2022, 16(1): 2-17. doi: 10.1093/ecco-jcc/jjab178
[4] Lamb CA, Kennedy NA, Raine T, et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults[J]. Gut, 2019, 68(Suppl 3): s1-s106. doi: 10.1136/gutjnl-2019-318484
[5] Hazel K, O'Connor A. Emerging treatments for inflammatory bowel disease[J]. Ther Adv Chronic Dis, 2020, 11: 2040622319899297.
[6] Little D, Tabatabavakili S, Shaffer SR, et al. Effectiveness of Dose De-escalation of Biologic Therapy in Inflammatory Bowel Disease: A Systematic Review[J]. Am J Gastroenterol, 2020, 115(11): 1768-1774. doi: 10.14309/ajg.0000000000000783
[7] Gisbert JP, Marín AC, Chaparro M. The Risk of Relapse after Anti-TNF Discontinuation in Inflammatory Bowel Disease: Systematic Review and Meta-Analysis[J]. Am J Gastroenterol, 2016, 111(5): 632-647. doi: 10.1038/ajg.2016.54
[8] 毛堂友, 程佳伟, 魏仕兵, 等. 清肠温中方治疗溃疡性结肠炎84例[J]. 环球中医药, 2016, 9(4): 479-481. https://www.cnki.com.cn/Article/CJFDTOTAL-HQZY201811057.htm
[9] 王志斌, 陈晨, 郭一, 等. 清肠温中方治疗轻中度溃疡性结肠炎的临床研究[J]. 中国中西医结合杂志, 2018, 38(1): 15-19. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZXJ201801004.htm
[10] 孙中美, 陈晓伟, 胡立明, 等. 清肠温中方对缓解期溃疡性结肠炎的远期作用研究[J]. 中国中西医结合消化杂志, 2021, 29(9): 619-623. http://zxpw.cbpt.cnki.net/WKD2/WebPublication/paperDigest.aspx?paperID=91bb364f-b931-4df9-981a-ea61ba6820d3
[11] Sun Z, Li J, Wang W, et al. Qingchang Wenzhong Decoction Accelerates Intestinal Mucosal Healing Through Modulation of Dysregulated Gut Microbiome, Intestinal Barrier and Immune Responses in Mice[J]. Front Pharmacol, 2021, 12: 738152. doi: 10.3389/fphar.2021.738152
[12] Sun Z, Li J, Dai Y, et al. Indigo Naturalis Alleviates Dextran Sulfate Sodium-Induced Colitis in Rats via Altering Gut Microbiota[J]. Front Microbiol, 2020, 11: 731. doi: 10.3389/fmicb.2020.00731
[13] 甄建华, 黄光瑞. 溃疡性结肠炎病因和发病机制的现代医学研究进展[J]. 世界华人消化杂志, 2019, 27(4): 245-251. https://www.cnki.com.cn/Article/CJFDTOTAL-XXHB201904006.htm
[14] Iwamuro M, Takahashi T, Watanabe N, et al. Enriched CD45RA-CD62 L+ central memory T and decreased CD3+CD56+ natural killer T lymphocyte subsets in the rectum of ulcerative colitis patients[J]. Int J Immunopathol Pharmacol, 2022, 36: 20587384211051982.
[15] Zundler S, Becker E, Spocinska M, et al. Author Correction: Hobit-and Blimp-1-driven CD4+ tissue-resident memory T cells control chronic intestinal inflammation[J]. Nat Immunol, 2019, 20(4): 514.
[16] Chen B, Ye B, Li M, et al. TIGIT Deficiency Protects Mice From DSS-Induced Colitis by Regulating IL-17A-Producing CD4+ Tissue-Resident Memory T Cells[J]. Front Immunol, 2022, 13: 931761.
[17] Geltink R, Kyle RL, Pearce EL. Unraveling the Complex Interplay Between T Cell Metabolism and Function[J]. Annu Rev Immunol, 2018, 36: 461-488.
[18] Pérez-Pérez A, Vilariño-García T, Fernández-Riejos P, et al. Role of leptin as a link between metabolism and the immune system[J]. Cytokine Growth Factor Rev, 2017, 35: 71-84.
[19] 陈晓伟, 高康丽, 李世荣, 等. 清肠温中方治疗轻中度溃疡性结肠炎56例[J]. 环球中医药, 2018, 11(11): 1826-1829. https://www.cnki.com.cn/Article/CJFDTOTAL-HQZY201811057.htm
-




 下载:
下载: