Clinical efficacy observation of Jidesheng snake medicine in the treatment of patients with middle and advanced liver cancer of damp-heat and stasis toxin syndrome and correlation study of its influence on serum miR-335
-
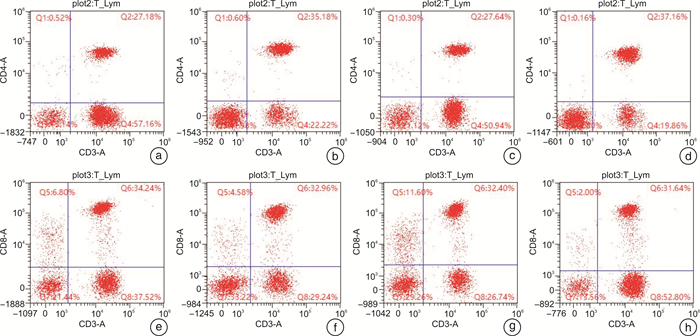
摘要: 目的 观察季德胜蛇药治疗中晚期湿热瘀毒型肝癌患者的临床疗效以及对患者血清miR-335的影响。方法 选取2019年1月—2021年9月在南通大学附属南通第三医院住院部准备接受治疗的62例符合研究条件的中晚期湿热瘀毒型肝癌患者,将他们随机分为观察组和对照组各31例,在西医对症药物治疗的基础上观察组给予季德胜蛇药口服治疗3个月,而对照组不予中药治疗,3个月后进行复诊。对比2组肝癌患者治疗前后的谷草转氨酶(AST)、谷丙转氨酶(ALT)、白蛋白(ALB)、总胆红素(TBIL)情况,治疗前后T淋巴细胞水平变化情况,加用季德胜蛇药治疗后的临床有效率以及患者生活质量改变情况,对比分析不良反应的发生率。比较治疗时、治疗后1个月、治疗后3个月患者外周血中miR-335表达水平。结果 治疗后,2组患者AST、ALT、TBIL均低于治疗前(P< 0.05),同时观察组这3项指标均显著低于对照组(P< 0.05);2组治疗前后ALB指标无明显变化(P>0.05)。治疗后,2组患者T淋巴细胞检测CD4+、CD4+/CD8+指标均较治疗前升高(P< 0.05),且观察组均高于对照组(P< 0.05);2组治疗前后CD8+表达水平无明显改变(P>0.05)。加用季德胜蛇药治疗后的肝癌患者临床治疗有效率为74.19%,高于对照组的61.29%(P< 0.05);生活质量提高率为67.74%,高于对照组的48.39%(P< 0.05);生活质量下降率为12.90%,低于对照组的25.81%(P< 0.05);加用季德胜蛇药治疗后没有提高药物不良反应发生率(P>0.05)。2组患者治疗时外周血miR-335表达差异无统计学意义(P>0.05),治疗后1个月观察组外周血miR-335表达较治疗时有所升高,治疗后3个月明显高于治疗时(P< 0.05),而对照组基本持平(P=0.969)。结论 应用季德胜蛇药对中晚期湿热瘀毒型肝癌患者可以提高患者临床疗效,改善患者生活质量,并且能提高患者外周血miR-335表达水平,具有一定的抗癌临床应用意义。Abstract: Objective To observe the clinical effect of Jidesheng snake medicine on patients with middle and advanced liver cancer of damp-heat and stasis toxin syndrome and study its effect on serum miR-335.Methods Sixty-two patients with middle and advanced liver cancer of damp-heat and stasis toxin syndrome who were prepared for basic treatment in the inpatient department of Nantong Third Hospital were selected from January 2019 to September 2021. They were randomly divided into observation group and control group(n=31), on the basis of western medicine symptomatic drug treatment, the observation group was given Jidesheng snake medicine oral treatment for 3 months, while the control group was not given Traditional Chinese Medicine treatment. A follow-up visit was made after 3 months. The levels of AST, ALT, ALB, TBIL and the changes of T lymphocyte level were compared between the two groups of liver cancer patients before and after treatment. To compare the clinical effective rate and life quality of patients treated with Jidesheng snake medicine. The incidence of adverse reactions was analyzed. The expression levels of miR-335 in peripheral blood of patients were compared at the time of treatment, one month and three months after treatment.Results AST, ALT and TBIL after treatment in two groups were lower than before treatment(P< 0.05), and the above three indexes in observation group were significantly lower than those in control group(P< 0.05). There was no significant change in ALB before and after treatment in both groups(P>0.05). CD4+ and CD4+/CD8+ indexes of T lymphocytes after treatment in two groups were increased compared with those before treatment(P< 0.05), and the two indexes in observation group were higher than those in control group(P< 0.05). There was no significant change in CD8+ expression before and after treatment(P>0.05). The effective rate of liver cancer patients treated with Jidesheng snake medicine was 74.19%, which was higher than that of 61.29% in the control group(P< 0.05), the improvement rate of life quality was 67.74%, which was higher than that of 48.39% in control group(P< 0.05), the reduction rate of quality of life was 12.90%, which was lower than that of 25.81% in the control group(P< 0.05). No increase in the incidence of adverse drug reactions after the addition of Jidesheng snake medicine treatment(P>0.05). There was no significant difference in the expression of miR-335 in peripheral blood between two groups(P>0.05), the expression of miR-335 in peripheral blood of the observation group was little increased after one month, and significantly higher after three months(P< 0.05), the control group was basically flat(P=0.969).Conclusion Jidesheng snake medicine can improve the clinical efficacy, improve the life quality of patients, and improve the expression level of miR-335 in peripheral blood in patients with middle and advanced liver cancer of damp-heat and stasis toxin syndrome, which has certain anticancer clinical application significance.
-
Key words:
- Jidesheng snake medicine /
- liver cancer /
- clinical efficacy /
- miR-335
-

-
表 1 2组患者的基线特征
项目 对照组
(n=31)观察组
(n=31)χ2/t P 年龄/岁 63.68±9.38 59.10±9.11 0.328 0.576 性别/例 0.075 0.785 男 20 19 女 11 12 肝癌病程/年 5.23±2.34 4.98±1.79 0.228 0.626 乙肝患者/例 27 29 0.127 0.814 Child-Pugh分级/例 0.042 0.839 A级 20 18 B级 11 13 治疗史/例 0.321 0.643 TACE术治疗 7 8 TACE术+系统治疗 24 23 表 2 2组患者治疗前后的肝功能指标变化
X±S 指标 对照组(n=31) 观察组(n=31) t P AST/(U·L-1) 治疗前 79.10±22.66 74.43±17.80 0.141 0.761 治疗后 53.00±14.06 42.33±2.78 2.782 0.044 t 2.987 5.231 P 0.034 <0.001 ALT/(U·L-1) 治疗前 104.50±19.41 100.31±20.26 0.113 0.921 治疗后 82.32±11.23 71.21±23.45 2.953 0.035 t 3.253 5.341 P 0.021 0.000 ALB/(g·L-1) 治疗前 31.34±3.23 30.23±2.35 0.063 0.872 治疗后 32.12±3.21 29.11±2.45 0.952 0.292 t 0.354 0.328 P 0.431 0.576 TBIL/(μmol·L-1) 治疗前 26.12±6.12 27.13±4.43 0.021 0.882 治疗后 22.15±4.23 19.21±4.23 4.132 0.023 t 3.185 5.968 P 0.025 0.000 表 3 2组患者治疗前后T淋巴细胞水平变化
X±S 指标 对照组(n=31) 观察组(n=31) t P CD4+/% 治疗前 27.32±4.31 27.53±4.32 0.124 0.821 治疗后 35.24±3.51 37.69±4.57 3.782 0.036 t 3.429 5.325 P 0.032 <0.001 CD8+/% 治疗前 34.23±2.81 32.70±2.96 0.073 0.091 治疗后 32.94±2.46 31.85±2.52 1.953 0.435 t 1.376 1.967 P 0.265 0.324 CD4+/CD8+ 治疗前 1.22±0.16 1.32±0.14 1.163 0.222 治疗后 1.52±0.39 1.78±0.54 3.153 0.032 t 3.448 3.476 P 0.032 0.026 表 4 2组患者临床治疗效果比较
例 组别 例数 疾病进展 疾病稳定 部分缓解 完全缓解 临床有效率/% 对照组 31 12 11 8 0 61.29 观察组 31 8 12 11 0 74.19 χ2 4.324 P 0.033 表 5 2组生活质量改善情况比较
例(%) 组别 例数 改善 稳定 下降 对照组 31 15(48.39) 8(25.81) 8(25.81) 观察组 31 21(67.74) 6(19.35) 4(12.90) χ2 5.034 0.884 4.592 P 0.025 0.347 0.033 表 6 2组患者治疗后的不良反应比较
例(%) 组别 例数 过敏 肾功能受损 胃肠道不适 神经受损 发生率/% 对照组 31 1(3.2) 2(6.5) 2(6.5) 0 16.1 观察组 31 0 1(3.2) 3(9.7) 1(3.2) 16.1 χ2 1.40 2.01 0.17 2.24 0.01 P 0.54 0.44 0.65 0.35 0.91 表 7 不同时间点2组患者外周血中miR-335的表达水平
X±S 时间点 对照组 观察组 t P 治疗时 1.23±0.18 1.27±0.26 0.145 0.885 治疗1个月后 1.28±0.25 1.45±0.16 0.561 0.576 治疗3个月后 1.31±0.21 2.11±0.28 2.258 0.028 F 0.032 3.302 P 0.969 0.041 -
[1] 李佳莹, 张秋丽, 张修礼. 射频消融在早期肝癌治疗中的应用进展[J]. 中华消化病与影像杂志(电子版), 2021, 11(5): 226-228. doi: 10.3877/cma.j.issn.2095-2015.2021.05.010
[2] 卢梅, 戴锴, 陈莎. 肝癌细胞免疫治疗研究进展[J]. 医学综述, 2021, 27(15): 2972-2976. doi: 10.3969/j.issn.1006-2084.2021.15.012
[3] 吴驻林, 魏春山, 康建媛, 等. 中医益气健脾解毒法辅助治疗原发性肝癌的荟萃分析[J]. 中医肿瘤学杂志, 2021, 3(4): 94-101. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYZL202104017.htm
[4] 倪雪娇, 许春明, 徐爱兵, 等. 季德胜蛇药联合化疗治疗原发性肝癌的临床效果观察[J]. 实用临床医药杂志, 2019, 23(24): 61-63. https://www.cnki.com.cn/Article/CJFDTOTAL-XYZL201924019.htm
[5] Liang H, Zhang C, Guan H, et al. LncRNA DANCR promotes cervical cancer progression by upregulating ROCK1 via sponging miR-335-5p[J]. J Cell Physiol, 2019, 234(5): 7266-7278. doi: 10.1002/jcp.27484
[6] Zhang P, Yang X, Wang L, et al. Overexpressing miR-335 inhibits DU145 cell proliferation by targeting early growth response 3 in prostate cancer[J]. Int J Oncol, 2019, 54(6): 1981-1994.
[7] 姚丽娜, 陈国忠, 张磊, 等. 血清外泌体中miR-335-5p作为诊断三阴性乳腺癌的生物标志物研究[J]. 中华内分泌外科杂志, 2021, 15(5): 458-462. doi: 10.3760/cma.j.cn.15807-20210508-00149
[8] 曹英, 马许辉, 张卉, 等. 联合检测HCC患者血清中GP73、GLYPICAN-3、MiR-335表达水平与患者诊断价值的关系[J]. 实验与检验医学, 2021, 39(3): 706-709. doi: 10.3969/j.issn.1674-1129.2021.03.060
[9] 杨栋勇, 徐源, 樊冀闽, 等. 抑制lncRNA LINC01503通过靶向调控miR-335-5p对肺癌细胞活力、迁移和侵袭的影响及其机制研究[J]. 中国病理生理杂志, 2020, 36(4): 619-627. doi: 10.3969/j.issn.1000-4718.2020.04.006
[10] 张玉, 陈琳, 卞兆连, 等. miR-335在肝细胞肝癌血清中的表达及其意义[J]. 中华肝脏病杂志, 2017, 25(1): 47-49. doi: 10.3760/cma.j.issn.1007-3418.2017.01.012
[11] 张玉, 邵建国, 陈琳, 等. 季德胜蛇药通过调控miR-335抑制肝癌Huh-7细胞增殖作用机制探讨[J]. 环球中医药, 2017, 10(4): 452-456. doi: 10.3969/j.issn.1674-1749.2017.04.015
[12] 中华人民共和国国家卫生健康委员会医政医管局. 《原发性肝癌诊疗规范(2019年版)》[J]. 中华肝脏病杂志, 2020, 28(2): 112-128. doi: 10.3760/cma.j.issn.1007-3418.2020.02.004
[13] 周仲瑛. 中医内科学[M]. 北京: 中国中医药出版社, 2014: 456.
[14] 张炫, 李晓辉. 肝动脉化疗栓塞联合射频消融治疗原发性肝癌的临床观察[J]. 解放军医药杂志, 2020, 32(5): 29-32. doi: 10.3969/j.issn.2095-140X.2020.05.007
[15] 蒋兆荣. 联合化疗治疗中晚期原发性肝癌的临床研究[J]. 中国实用医药, 2019, 14(36): 124-126. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSSA201936067.htm
[16] 张超, 黄喜燕, 李祥, 等. 重楼皂苷Ⅶ对肝癌的体内抗肿瘤作用及机制研究[J]. 中药新药与临床药理, 2022, 33(1): 7-13. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYXY202201002.htm
[17] 滕永杰, 柳卓, 廖柳, 等. 蜈蚣提取物对人肝癌细胞HepG2及肝癌原位移植瘤STAT3信号通路的影响[J]. 数字中医药(英文), 2020, 3(2): 67-79. https://www.cnki.com.cn/Article/CJFDTOTAL-DFGF202002002.htm
[18] 刘薰, 杨柱, 龙奉玺, 等. 蟾蜍等有毒中药在肿瘤防治中的应用[J]. 西部中医药, 2020, 33(9): 129-132. https://www.cnki.com.cn/Article/CJFDTOTAL-GSZY202009037.htm
[19] 王耀杰, 相晓晗, 韩丽娜, 等. 中药地锦草乙醇提取物体外抑制胃癌细胞增殖活性及机制研究[J]. 癌变·畸变·突变, 2020, 32(3): 187-193. https://www.cnki.com.cn/Article/CJFDTOTAL-ABJB202003006.htm
[20] 李慧, 姚建华, 田芝奥, 等. 季德胜蛇药含药血清对人肝癌细胞Hep-G2增殖和凋亡的影响[J]. 中西医结合肝病杂志, 2012, 22(1): 32-33, 37. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXGB201201014.htm
[21] 姚建华, 田芝奥, 李慧, 等. 季德胜蛇药联合TACE治疗中晚期肝癌的临床观察[J]. 实用肝脏病杂志, 2011, 14(5): 379-380. https://www.cnki.com.cn/Article/CJFDTOTAL-GBSY201105031.htm
[22] 沈宗毅, 李卯晨, 白素杭, 等. 肝癌免疫治疗的研究进展[J]. 生物工程学报, 2019, 35(12): 2326-2338. https://www.cnki.com.cn/Article/CJFDTOTAL-SHWU201912011.htm
[23] 邓永, 唐腾骞, 张雷达. miRNA-200b逆转肝细胞癌HepG2细胞对索拉菲尼的耐药性及其相关机制[J]. 第三军医大学学报, 2021, 43(22): 2435-2440. https://www.cnki.com.cn/Article/CJFDTOTAL-DSDX202122005.htm
[24] 彭契六, 韦尚谋, 张磊, 等. miRNA-152在肝癌组织中的表达及临床意义[J]. 肿瘤防治研究, 2021, 48(8): 769-773. https://www.cnki.com.cn/Article/CJFDTOTAL-ZLFY202108005.htm
[25] 王汇锋, 陈洁, 赵咫龙. miR-4282对肝癌细胞侵袭、迁移能力的影响[J]. 锦州医科大学学报, 2021, 42(4): 15-20. https://www.cnki.com.cn/Article/CJFDTOTAL-JZYX202104004.htm
[26] 刘雪婷, 王莹, 宋家羽, 等. miR-335对恶性肿瘤的调控作用及其临床应用的研究进展[J]. 现代肿瘤医学, 2022, 30(4): 721-724. https://www.cnki.com.cn/Article/CJFDTOTAL-SXZL202204035.htm
[27] Li HW, Liu J. Circ_0009910 promotes proliferation and metastasis of hepato-cellular carcinoma cells through miR-335-5p/ROCK1 axis[J]. Eur Rev Med Pharmacol Sci, 2020, 24: 1725-1735.
[28] 张小舟, 帅领, 王庆大, 等. 接受TACE治疗的肝细胞癌患者血清miR-335水平变化及其对预后判断的价值分析[J]. 实用肝脏病杂志, 2019, 22(2): 268-271. https://www.cnki.com.cn/Article/CJFDTOTAL-GBSY201902029.htm
-




 下载:
下载: