Correlation analysis between thyroid stimulating hormone and liver function and prognosis in chronic severe hepatitis patients
-
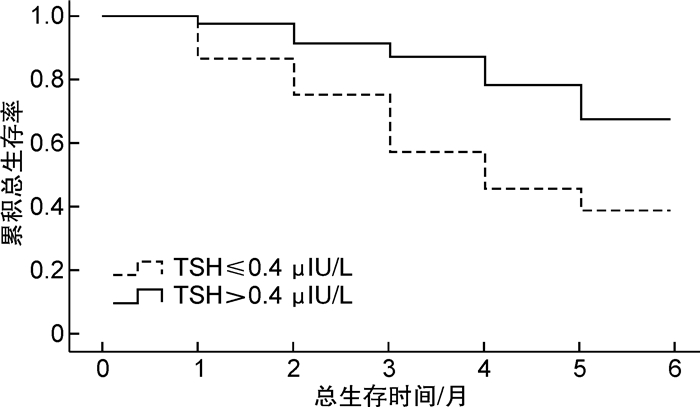
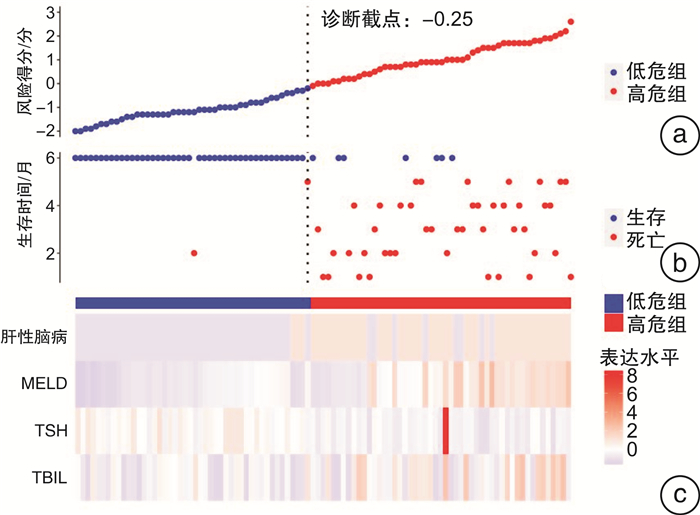
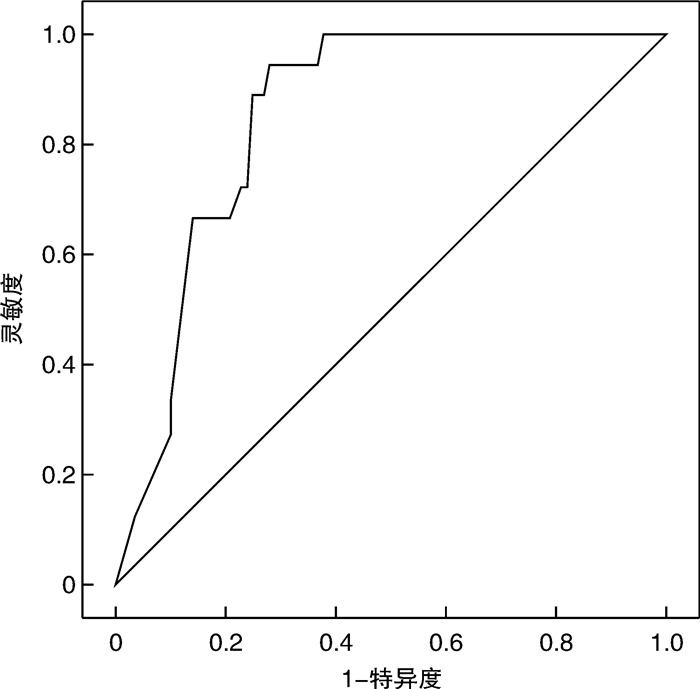
摘要: 目的 探讨慢性重型肝炎患者促甲状腺激素(thyroid stimulating hormone,TSH)水平与肝功能以及预后的相关性。方法 选取2020年1月—2022年4月就诊的慢性重型肝炎患者为研究对象,所有患者出院后均随访6个月,以随访6个月内患者生存状态分为存活组(52例)和死亡组(45例),比较两组患者的临床资料以及血清TSH表达水平,Cox回归分析获得影响慢性重型肝炎患者随访6个月死亡的独立预测因素,绘制TSH预测慢性重型肝炎患者随访6个月死亡的ROC曲线,以TSH的最佳诊断截点将所有患者分为两组,Kaplan-Meier生存分析比较两组患者随访6个月的总生存率,并基于TSH和其他独立预测因素绘制相关Cox死亡风险得分图,Pearson相关性分析探讨TSH与肝功能的相关性。结果 死亡组患者肝性脑病、肝肾综合征所占比例、总胆红素以及终末期肝病模型(model for end-stage liver disease,MELD)评分明显高于存活组,凝血酶原活动度(prothrombin activity,PTA)和TSH明显低于存活组(P < 0.05)。多因素Cox回归分析显示,肝性脑病、总胆红素、MELD评分以及TSH为影响慢性重型肝炎患者随访6个月死亡的独立预测因素(P < 0.05)。ROC分析结果显示,TSH预测慢性重型肝炎患者随访6个月内死亡的曲线下面积为0.843(95%CI:0.745~0.912),最佳诊断截点为0.4 μIU/L。Kaplan-Meier生存分析显示,TSH≤0.4 μIU/L组患者随访6个月总生存率(37.5%)明显低于TSH>0.4 μIU/L组(64.9%),差异有统计学意义(P < 0.05)。Cox死亡风险得分模型显示,患者血清TSH表达水平与Cox死亡风险得分呈负相关,与总生存时间呈正相关。相关性分析显示,慢性重型肝炎患者TSH与谷草转氨酶、谷丙转氨酶、白蛋白以及PTA水平呈正相关(P < 0.05),与总胆红素、MELD评分以及肝性脑病呈负相关(P < 0.05)。结论 TSH表达水平越低,慢性重型肝炎患者肝功能越差,肝性脑病发生率越高,生存率越低。Abstract: Objective To explore the correlation between thyroid stimulating hormone(TSH) and liver function and prognosis in chronic severe hepatitis patients.Methods One hundred and eleven chronic severe hepatitis patients who visited our hospital from January 2020 to April 2022 were selected. All patients were followed up for 6 months after discharge. The patients were divided into survival group(52 cases) and death group(45 cases) according to their survival status within six months follow-up. The general clinical data and serum TSH expression level of patients between the two groups were compared. Cox regression analysis was used to obtain independent predictors of the death in patients who were followed-up for six months. ROC curve of TSH predicting the death of chronic severe hepatitis patients within six months follow-up was drawn. All patients were divided into two groups according to the best diagnostic cutoff point of TSH. Kaplan-Meier survival analysis was uesed to compare the 6-month overall survival rate of patients between the two groups, and the relevant Cox death risk score chart based on TSH and other independent predictors was drawn. Pearson correlation analysis was used to explore the correlation between TSH and liver function.Results The proportion of hepatic encephalopathy, hepatorenal syndrome, total bilirubin and model for end-stage liver disease(MELD) score of patients in death group were significantly higher than those in survival group, while prothrombin activity(PTA) and TSH were significantly lower than those in survival group(P < 0.05). Multivariate Cox regression analysis showed that hepatic encephalopathy, total bilirubin, MELD score and TSH were independent predictors of death in chronic severe hepatitis patients within 6 months follow-up(P < 0.05). ROC analysis results showed that the area under the curve(AUC) of TSH predicting death in chronic severe hepatitis patients within six months follow-up was 0.843(95%CI: 0.745-0.912), and the best cut-off point of TSH was 0.4 μIU/L. Kaplan Meier survival analysis showed that the six-month overall survival rate(37.5%) of patients in TSH ≤ 0.4 μIU/L group was significantly lower than that in TSH>0.4 μIU/L group(64.9%)(P < 0.05). The Cox death risk score model showed that the serum TSH expression level was negatively correlated with the Cox death risk score, and positively correlated with the total survival time. Correlation analysis showed that TSH was positively correlated with AST, ALT, albumin and PTA in chronic severe hepatitis patients(P < 0.05), and negatively correlated with total bilirubin, MELD score and hepatic encephalopathy(P < 0.05).Conclusion The lower the expression level of TSH, the worse the liver function of chronic severe hepatitis patients, the higher the incidence of hepatic encephalopathy, and the lower the survival rate.
-
Key words:
- thyroid stimulating hormone /
- chronic severe hepatitis /
- liver function /
- prognosis
-

-
表 1 两组患者的临床资料以及血清TSH表达水平比较
X±S,例,M(P25,P75) 临床资料 存活组(n=52) 死亡组(n=45) t/Z/χ2 P 年龄/岁 53.2±7.6 53.8±7.3 0.145 0.876 性别 1.435 0.145 男 36 35 女 16 10 病因 1.228 0.184 乙肝 30 22 丙肝 12 10 其他 10 13 食管胃底静脉曲张程度 0.986 0.278 中度 34 22 重度 18 23 肝性脑病 9.229 < 0.001 是 10 34 否 42 11 肝肾综合征 5.112 0.025 是 12 23 否 40 22 消化道出血 1.345 0.169 是 20 20 否 32 25 AST/(U/L) 51.4±8.9 49.2±9.2 0.678 0.412 ALT/(U/L) 53.9±7.8 50.2±8.3 0.784 0.334 白蛋白/(g/L) 26.3±4.8 25.5±5.5 0.287 0.765 TBIL/(mmol/L) 311.2±8.9 376.4±11.2 9.566 < 0.001 PTA/% 35.4±4.8 28.2±5.7 6.887 < 0.001 MELD评分/分 24.3±5.9 38.5±6.2 8.345 < 0.001 甲胎蛋白/(μg/L) 123.5±9.7 118.5±9.6 1.014 0.189 CRP/(mg/L) 3.2(2.3, 3.9) 3.4(2.4, 4.0) 0.568 0.487 PCT/(ng/L) 53.8(45.6, 63.4) 55.7(46.7, 65.9) 0.439 0.569 TSH/(μIU/L) 1.1±0.3 0.3±0.1 4.588 < 0.001 表 2 慢性重型肝炎患者随访6个月死亡的单因素Cox回归分析
自变量 β SE Waldχ2 P HR 95% CI 肝性脑病 1.678 0.337 13.677 < 0.001 5.336 1.563~9.663 TBIL 0.832 0.118 11.221 < 0.001 2.568 1.135~4.869 PTA -0.789 0.113 5.423 0.027 0.517 0.238~0.823 肝肾综合征 1.274 0.356 5.875 0.021 4.567 1.768~8.774 MELD评分 1.367 0.127 12.234 < 0.001 4.945 1.078~4.889 TSH -0.894 0.238 10.988 < 0.001 0.459 0.248~0.865 表 3 慢性重型肝炎患者随访6个月死亡的多因素Cox回归分析
自变量 β SE Waldχ2 P HR 95% CI 肝性脑病 1.378 0.412 15.894 < 0.001 5.036 1.369~9.894 TBIL 0.783 0.218 10.234 0.001 2.369 1.156~4.547 MELD评分 1.292 0.432 9.238 0.003 4.846 1.029~8.435 TSH -0.812 0.339 7.487 0.010 0.485 0.212~0.738 表 4 慢性重型肝炎患者TSH与肝功能的相关性分析
指标 AST ALT 白蛋白 TBIL PTA MELD评分 肝性脑病 r 0.578 0.554 0.654 -0.689 0.549 -0.692 -0.672 P 0.018 0.031 < 0.001 < 0.001 0.027 < 0.001 < 0.001 -
[1] 唐弋均, 秦学斌, 何凡, 等. 组合型非生物型人工肝治疗慢性重型肝炎的临床疗效及患者长期预后[J]. 中国中西医结合消化杂志, 2022, 30(6): 407-410, 418. http://zxpw.cbpt.cnki.net/WKD2/WebPublication/paperDigest.aspx?paperID=aef34b03-0563-4a4c-9a3a-0dd7b80ce0dd
[2] 刘晨帆, 李建志, 张照华, 等. 慢性重型肝炎患者肝脏血流动力学改变及临床意义[J]. 系统医学, 2019, 4(4): 9-11, 23. https://www.cnki.com.cn/Article/CJFDTOTAL-XTYX201904004.htm
[3] de Sanctis V, Elsedfy H, Soliman AT, et al. Acquired hypogonadotropic hypogonadism(AHH)in thalassaemia major patients: an underdiagnosed condition?[J]. Mediterr J Hematol Infect Dis, 2016, 8(1): e2016001.
[4] 赵阳. 还原型谷胱甘肽联合促肝细胞生长素治疗慢性重型肝炎的临床疗效[J]. 实用中西医结合临床, 2021, 21(5): 11-12. https://www.cnki.com.cn/Article/CJFDTOTAL-SZXL202105005.htm
[5] 柯比努尔·吐尔逊, 韩丹, 张跃新. 肝硬化患者血清促甲状腺激素和甲状腺素水平变化及其临床意义[J]. 实用肝脏病杂志, 2017, 20(6): 764-766. https://www.cnki.com.cn/Article/CJFDTOTAL-GBSY201706034.htm
[6] Attard CC, Sze WCC, Vella S. Predictors of autoimmune thyroid disease[J]. Proc(Bayl Univ Med Cent), 2022, 35(5): 608-614.
[7] 马景胜, 杨文涛, 朱红飞, 等. 甲状腺激素检测在慢性重型肝炎患者肝移植围手术期的意义[J]. 中国中西医结合消化杂志, 2022, 30(1): 42-44. http://zxpw.cbpt.cnki.net/WKD2/WebPublication/paperDigest.aspx?paperID=2df98ee5-ed21-4e30-955d-74bc87961f2d
[8] 中华医学会感染病学分会肝衰竭与人工肝学组, 中华医学会肝病学分会重型肝病与人工肝学组. 肝衰竭诊治指南(2018年版)[J]. 中华临床感染病杂志, 2018, 11(6): 401-410. doi: 10.3760/cma.j.issn.1674-2397.2018.06.001
[9] 张丽, 武希润. 肝硬化患者血清甲状腺激素水平变化的临床意义分析[J]. 临床医药文献电子杂志, 2018, 5(15): 75-78. https://www.cnki.com.cn/Article/CJFDTOTAL-LCWX201815059.htm
[10] 吴文文, 陈永忠, 仝亚林. 肝硬化患者血清甲状腺激素水平与肝硬化严重程度的关系[J]. 中国实用医刊, 2019, 46(22): 34-37.
[11] Guo W, Qin P, Li XN, et al. Free triiodothyronine is associated with hepatic steatosis and liver stiffness in euthyroid Chinese adults with non-alcoholic fatty liver disease[J]. Front Endocrinol(Lausanne), 2021, 12: 711956. doi: 10.3389/fendo.2021.711956
[12] Wang XL, Mao JB, Zhou XL, et al. Thyroid stimulating hormone triggers hepatic mitochondrial stress through cyclophilin D acetylation[J]. Oxid Med Cell Longev, 2020, 2020: 1249630.
[13] Naguib R, Fayed A, Elkemary EZ, et al. Evaluation of thyroid function and thyroid autoimmune disease in patients with non-alcoholic fatty liver disease[J]. Clin Exp Hepatol, 2021, 7(4): 422-428. doi: 10.5114/ceh.2021.111169
[14] Zhou Z, Xu MX, Lin N, et al. Associating liver partition and portal vein ligation for staged hepatectomy versus conventional two-stage hepatectomy: a systematic review and meta-analysis[J]. World J Surg Oncol, 2017, 15(1): 227.
[15] Patel AM, Stanback C, Vellanki P. Clinical case report: dissociation of clinical course of coexisting autoimmune hepatitis and Graves disease[J]. AACE Clin Case Rep, 2021, 7(1): 36-39.
[16] Babic Leko M, Gunjaca I, Pleic N, et al. Environmental factors affecting thyroid-stimulating hormone and thyroid hormone levels[J]. Int J Mol Sci, 2021, 22(12): 6521.
[17] Liu YP, Yang HY, Liang CF, et al. Expression of functional thyroid-stimulating hormone receptor in microglia[J]. Ann Endocrinol(Paris), 2022, 83(1): 40-45.
[18] D'Ambrosio R, Campi I, Maggioni M, et al. The relationship between liver histology and thyroid function tests in patients with non-alcoholic fatty liver disease(NAFLD)[J]. PLoS One, 2021, 16(4): e0249614.
[19] Nakamura Y, Yoshihara A, Kiriya M, et al. Thyroid stimulating hormone suppresses the expression and activity of cytosolic sulfotransferase 1a1 in thyrocytes[J]. Endocr J, 2022, 69(10): 1261-1269.
[20] Yi FM, Zhang W, Feng L. Efficacy and safety of different options for liver regeneration of future liver remnant in patients with liver malignancies: a systematic review and network meta-analysis[J]. World J Surg Oncol, 2022, 20(1): 399.
[21] Mammen JS, Ghazarian SR, Pulkstenis E, et al. Phenotypes of interferon-α-induced thyroid dysfunction among patients treated for hepatitis C are associated with pretreatment serum TSH and female sex[J]. J Clin Endocrinol Metab, 2012, 97(9): 3270-3276.
[22] Hasselgren K, Rosok BI, Larsen PN, et al. Response to the comment on "ALPPS improves survival compared with TSH in patients affected of CRLM: survival analysis from the randomized controlled trial LIGRO-metastatic tumor burden in the future liver remnant for decision-making of staged hepatectomy"[J]. Ann Surg, 2021, 274(6): e750-e751.
[23] Chan KM, Wang YC, Wu TH, et al. Comment on "ALPPS improves survival compared with TSH in patients affected of CRLM survival analysis from the randomized controlled trial LIGRO": metastatic tumor burden in the future liver remnant for decision-making of staged hepatectomy[J]. Ann Surg, 2021, 274(6): e749-e750.
[24] Huang B, Wen WJ, Ye SD. TSH-SPP1/TRβ-TSH positive feedback loop mediates fat deposition of hepatocyte: Crosstalk between thyroid and liver[J]. Front Immunol, 2022, 13: 1009912.
[25] 高秀莹, 郭彩红, 朱巍, 等. 急性肝衰竭相关A型肝性脑病患者促甲状腺激素的异常及其相关影响因素[J]. 临床和实验医学杂志, 2019, 18(3): 274-277. https://www.cnki.com.cn/Article/CJFDTOTAL-SYLC201903014.htm
[26] Chen JF, Weng WZ, Huang M, et al. The impact of serum thyroid-stimulation hormone levels on the outcome of hepatitis B virus related acute-on-chronic liver failure: an observational study[J]. BMC Gastroenterol, 2022, 22(1): 330.
[27] Andacoglu O, Ozkan O, Sekmen U, et al. A review on the concepts for initially unresectable liver tumors-bridge to surgery[J]. Chirurgia(Bucur), 2022, 117(6): 635-642.
[28] Díaz Vico T, Granero Castro P, Alcover Navarro L, et al. Two stage hepatectomy(TSH)versus ALPPS for initially unresectable colorectal liver metastases: a systematic review and meta-analysis[J]. Eur J Surg Oncol, 2023, 49(3): 550-559.
[29] Wu YC, You SL, Zang H, et al. Usefulness of serum thyroid-stimulation hormone(TSH)as a prognostic indicator for acute-on-chronic liver failure[J]. Ann Hepatol, 2015, 14(2): 218-224.
[30] Bebars GM, Sayed MA, Hamdy L, et al. Effect of acute and chronic liver diseases on the thyroid function in children[J]. BMC Pediatr, 2021, 21(1): 361.
[31] Tozzoli R, Villalta D, Bizzaro N. Challenges in the standardization of autoantibody testing: a comprehensive review[J]. Clin Rev Allergy Immunol, 2017, 53(1): 68-77.
-




 下载:
下载: