Evaluation value of serum HIF-1α in disease activity and mucosal healing in ulcerative colitis patients
-
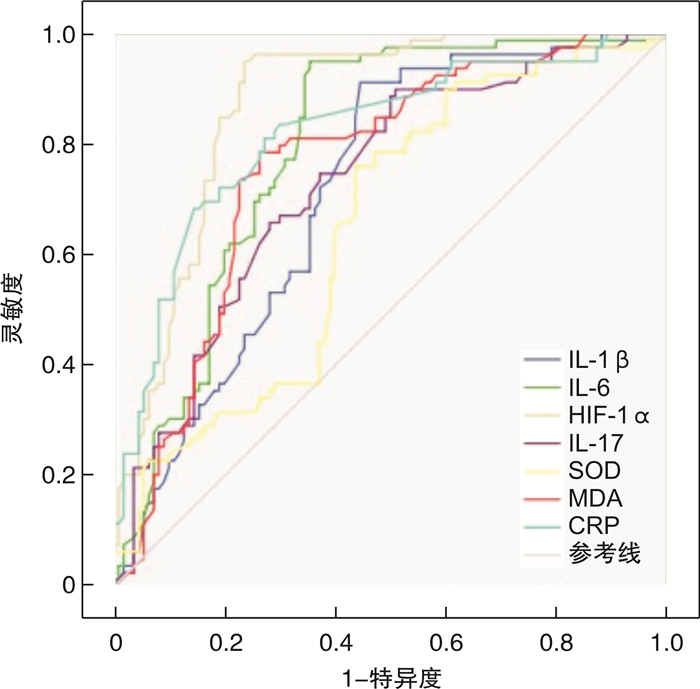
摘要: 目的 探讨血清低氧诱导因子-1α(HIF-1α)表达与溃疡性结肠炎(UC)疾病活动性及其黏膜愈合的关系。方法 以2017年9月—2022年3月收治的82例UC患者及30例同期健康体检者(对照组)为研究对象。采用ELISA法测定血清HIF-1α、IL-1β、IL-6、IL-17、C反应蛋白(CRP)水平,氯化硝基四氮唑蓝(NBT)法检测超氧化物歧化酶(SOD)含量,硫代巴比妥酸(TBA)法检测丙二醛(MDA)含量;分析HIF-1α表达与UC患者病情活动性、内镜分级、上述各指标及临床疗效的关系;采用受试者工作特征曲线(ROC)评估HIF-1α、炎症因子、CRP、氧化应激指标对内镜下黏膜愈合的预测价值。结果 活动期组UC患者血清HIF-1α水平较缓解期组和对照组明显升高,且HIF-1α在不同临床严重程度、内镜表现分级间表达差异有统计学意义,并与其呈显著正相关(r=0.812、0.719,P<0.05);随着UC临床严重程度增加,IL-1β、IL-6、IL-17及CRP水平增加,HIF-1α表达与其呈正相关(r=0.527、0.476、0.612、0.571,P<0.05);SOD表达下降,HIF-1α表达与其呈负相关(r=-0.497,P<0.05);MDA表达增加,HIF-1α表达与其呈正相关(r=0.502,P<0.05);ROC曲线结果显示,SOD、MDA、IL-1β、IL-17、IL-6、CRP、HIF-1α预测的曲线下面积(AUC)分别为0.648、0.761、0.718、0.731、0.795、0.804、0.874,具有一定的区分度。HIF-1α的AUC大于其他指标预测的AUC,区分度更好。HIF-1α在UC患者黏膜愈合预测中具有较高的灵敏度和特异度。结论 血清HIF-1α水平与UC疾病活动性及黏膜愈合具有相关性,血清HIF-1α表达水平随UC严重程度增加而增加,并且HIF-1α表达量与机体炎症反应、氧化应激反应功能紊乱程度具有相关性。血清HIF-1α在UC患者疾病活动性及黏膜愈合评估上具有重要意义。Abstract: Objective To investigate the relationship between the expression of serum hypoxia inducible factor-1 α (HIF-1 α) and the disease activity and mucosal healing in ulcerative colitis (UC).Methods Eighty-two patients with UC and 30 healthy subjects in the same period (control group) were admitted from September 2017 to March 2022. Determination of serum HIF-1α, IL-1β, IL-6, IL-17 and C-reactive protein (CRP) by ELISA. Determination of superoxide dismutase (SOD) by nitroblue tetrazolium (NBT) method. Determination of malondialdehyde (MDA) by thiobarbituric acid (TBA). To analyze the relationship between the expression of HIF-1α and disease activity, endoscopic grading, above-mentioned indexes and clinical efficacy in patients with UC. The value of HIF-1α, inflammatory cytokines, CRP and oxidative stress in predicting endoscopic mucosal healing was evaluated by receiver operating characteristic (ROC) curve.Results The level of serum HIF-1α in active UC patients was significantly higher than that in remission group and control group, and the expression of HIF-1α was significantly different among different clinical severity and endoscopic manifestation grades, and positively correlated with it (r=0.812, 0.719, P < 0.05). With the increase of clinical severity of UC, the levels of IL-1β, IL-6, IL-17 and CRP increased, and the expression of HIF-1α was positively correlated with it (r=0.527, 0.476, 0.612, 0.571, P < 0.05)); the expression of SOD decreased, and the expression of HIF-1α negatively correlated with it (r=-0.497, P < 0.05); the expression of MDA increased, and the expression of HIF-1α positively correlated with it(r=0.502, P < 0.05). The results of ROC curve show that the area under the curve (AUC) predicted by SOD, MDA, IL-1β, IL-17, IL-6, CRP and HIF-1α is 0.648, 0.761, 0.718, 0.731, 0.795, 0.804 and 0.874 respectively, which has a certain degree of distinction. The AUC of HIF-1α is higher than that of AUC predicted by other indexes, and the degree of differentiation is better. HIF-1α has high sensitivity and specificity in predicting mucosal healing in patients with UC.Conclusion The level of serum HIF-1α was correlated with the activity of UC disease and mucosal healing, and increased with the increase of the severity of UC, and the expression of HIF-1α was correlated with the degree of inflammation and oxidative stress. Serum HIF-1α plays an important role in the evaluation of disease activity and mucosal healing in patients with UC.
-

-
表 1 活动期UC患者血清HIF-1α表达水平与病情严重程度、内镜分级的相关性分析
X±S 指标 例数 HIF-1α/(ng·L-1) r P 临床病情 轻型 26 39.79±4.32 中型 21 70.35±4.231) 0.812 <0.05 重型 12 95.52±9.191)2) 内镜分级 Ⅰ级 23 38.79±3.50 Ⅱ级 26 68.87±9.693) 0.719 <0.05 Ⅲ级 10 97.50±8.753)4) 与轻型比较,1)P<0.05;与中型比较,2)P<0.05;与Ⅰ级比较,3)P<0.05;与Ⅱ级比较,4)P<0.05。 表 2 血清炎症因子及CRP的表达水平与患者病情严重程度的关系
pg/mL,X±S 病情 例数 IL-1β IL-6 IL-17 CRP 轻型 26 8.31±0.45 5.32±0.34 13.35±1.04 5.27±0.27 中型 21 15.11±0.431) 7.56±0.351) 20.81±2.031) 9.16±0.281) 重型 12 24.41±0.531)2) 13.12±0.921)2) 35.72±1.611)2) 13.22±0.321)2) 与轻型比较,1)P<0.05;与中型比较,2)P<0.05。 表 3 血清氧化应激指标的表达与患者病情严重程度的关系
X±S 病情 例数 SOD/(IU·mL-1) MDA/(μmol·L-1) 轻型 26 52.38±1.32 13.52±1.23 中型 21 35.61±1.161) 16.51±1.211) 重型 12 20.74±1.211)2) 22.92±1.171)2) 与轻型比较,1)P<0.05;与中型比较,2)P<0.05。 表 4 血清HIF-1α、炎症因子、CRP、氧化应激指标对UC患者内镜下黏膜愈合的预测价值
指标 AUC 截断值 灵敏度 特异度 阳性预测值/% 阴性预测值/% SOD 0.648 31.37 0.689 0.710 65.3 71.6 MDA 0.761 17.65 0.784 0.803 68.8 78.5 IL-1β 0.718 14.31 0.726 0.741 70.6 73.4 IL-17 0.731 22.37 0.715 0.782 71.4 72.3 IL-6 0.795 8.32 0.789 0.796 71.3 79.5 CRP 0.804 9.60 0.854 0.797 74.7 80.7 HIF-1α 0.874 72.69 0.873 0.836 77.5 83.6 表 5 不同病情严重程度UC患者入院、出院HIF-1α表达的比较
ng/L,X±S 病情 例数 入院HIF-1α 出院HIF-1α 轻型 26 42.07±12.88 29.64±9.421) 中型 21 71.93±16.45 41.26±13.651) 重型 12 86.63±5.98 56.74±9.571) 与入院HIF-1α比较,1)P<0.05。 -
[1] Marin M, Alzueta N, Pio M, et al. Ulcerative colitis induced by ixekizumab: a case report[J]. Eur J Hosp Pharm, 2021, 28(1): 50-52. doi: 10.1136/ejhpharm-2019-002016
[2] Greuter T, Vavricka SR. Extraintestinal manifestations in inflammatory bowel disease-epidemiology, genetics, and pathogenesis[J]. Expert Rev Gastroenterol Hepatol, 2019, 13(4): 307-317. doi: 10.1080/17474124.2019.1574569
[3] 岳宁宁, 陈香宇. VitD联合美沙拉秦治疗轻中度溃疡性结肠炎的临床疗效研究[J]. 医药论坛杂志, 2020, 41(12): 50-53. https://www.cnki.com.cn/Article/CJFDTOTAL-HYYX202012013.htm
[4] Yang M, Yang XM, Yin DH, et al. Beclin1 enhances cisplatin-induced apoptosis via Bcl-2-modulated autophagy in laryngeal carcinoma cells Hep-2[J]. Neoplasma, 2018, 65(1): 42-48. doi: 10.4149/neo_2018_161102N528
[5] 张静文. 溃疡性结肠炎内镜下评分与细胞免疫指标的相关性分析[J]. 实用医药杂志, 2020, 37(12): 1085-1087, 1089. doi: 10.14172/j.issn1671-4008.2020.12.009
[6] Novianti T, Juniantito V, Jusuf AA, et al. Expression and role of HIF-1αand HIF-2αin tissue regeneration: a study of hypoxia in house gecko tail regeneration[J]. Organogenesis, 2019, 15(3): 69-84. doi: 10.1080/15476278.2019.1644889
[7] Li H, Jia Y, Wang Y. Targeting HIF-1alpha signaling pathway for gastric cancer treatment[J]. Pharmazie, 2019, 74(1): 3-7.
[8] 冯力, 苏然. COPD患者血清8-isoprostane、HIF-1α、SP-D水平与肺功能及生活质量的相关性研究[J]. 国际检验医学杂志, 2021, 42(2): 214-217, 221. doi: 10.3969/j.issn.1673-4130.2021.02.020
[9] Malkov MI, Lee CT, Taylor CT. Regulation of the Hypoxia-Inducible Factor (HIF) by Pro-Inflammatory Cytokines[J]. Cells, 2021, 10(9): 2340-2354. doi: 10.3390/cells10092340
[10] 吴晌犊. 常美安配合美沙拉嗪对溃疡性结肠炎患者结肠黏膜ZO-1 mRNA、Occludin mRNA及T细胞亚群比例的影响[J]. 中国医师杂志, 2018, 20(5): 768-771. doi: 10.3760/cma.j.issn.1008-1372.2018.05.041
[11] 成斌. HIF-1α和COX-2在溃疡性结肠炎患者外周血及黏膜组织中的表达及临床意义[J]. 中国当代医药, 2020, 27(16): 4-6, 18. doi: 10.3969/j.issn.1674-4721.2020.16.003
[12] Yu SX, Li B, Hao JH, et al. Correlation of Hypoxia-inducible facto-1alpha and C-reactive protein with disease evaluation in patients with ulcerative colitis[J]. Am J Transl Res, 2020, 12(12): 7826-7835.
[13] Bertani L, Rossari F, Barberio B, et al. Novel prognostic biomarkers of mucosal healing in ulcerative colitis patients treated with anti-TNF: Neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio[J]. Inflamm Bowel Dis, 2020, 26(10): 1579-1587. doi: 10.1093/ibd/izaa062
[14] 宋泽军, 张明君, 任渝棠, 等. 改良Mayo内镜评分对溃疡性结肠炎有较高的评估价值[J]. 南方医科大学学报, 2022, 42(7): 997-1005. https://www.cnki.com.cn/Article/CJFDTOTAL-DYJD202207013.htm
[15] Cortes X, Borras-Blasco J, Flor L, et al. Successful use of golimumab in a patient with ulcerative colitis refractory to infliximab and adalimumab[J]. Int J Clin Pharmacol Ther, 2021, 59(3): 257-260. doi: 10.5414/CP202621
[16] Di Ruscio M, Variola A, Vernia F, et al. Role of ulcerative colitis endoscopic index of severity(UCEIS)versus Mayo endoscopic subscore(MES)in predicting patients' response to biological therapy and the need for colectomy[J]. Digestion, 2021, 102(4): 534-545. doi: 10.1159/000509512
[17] 周升凤, 张广清, 丁美祝, 等. 中西医结合联合慢病管理模式治疗溃疡性结肠炎患者的临床研究[J]. 世界中西医结合杂志, 2021, 16(12): 2304-2307. https://www.cnki.com.cn/Article/CJFDTOTAL-SJZX202112029.htm
[18] Shao T, Zhao CQ, Li FY, et al. Intestinal HIF-1α deletion exacerbates alcoholic liver disease by inducing intestinal dysbiosis and barrier dysfunction[J]. J Hepatol, 2018, 69(4): 886-895. doi: 10.1016/j.jhep.2018.05.021
[19] 郁丽亚, 缪敏慧, 尤建良. 溃疡性结肠炎患者血清HIF-1α、Beclin1、TLR4水平与其病情严重程度的关系研究[J]. 中国中西医结合消化杂志, 2020, 28(3): 206-209, 214. http://zxpw.cbpt.cnki.net/WKD2/WebPublication/paperDigest.aspx?paperID=665c4cac-4125-4038-8616-6b397bb71225
[20] Brown E, Rowan C, Strowitzki MJ, et al. Mucosal inflammation downregulates PHD1 expression promoting a barrier-protective HIF-1α response in ulcerative colitis patients[J]. FASEB J, 2020, 34(3): 3732-3742.
[21] Zamora C, Canto E, Nieto JC, et al. Inverse association between circulating monocyte-plarelet complexes and inflammation in ulcerative colitis patients[J]. Inflamm Bowel Dis, 2018, 24(4): 818-828.
[22] Angelidou I, Chrysanthopoulou A, Mitsios A, et al. REDD1/antophagy pathway is associated with neutrophil-driven IL-1β inflammatory response in active ulcerative colitis[J]. J Immunol, 2018, 200(12): 3950-3961.
[23] Kumar R, Singh AK, Starokadomskyy P, et al. Cutting edge: Hypoxia-induced Ubc9 promoter hypermethylation regulates IL-17 expression in ulcerative colitis[J]. J Immunol, 2021, 206(5): 936-940.
[24] 赵新跃. 美沙拉嗪联合双歧杆菌三联活菌对溃疡性结肠炎患者肠黏膜及机体氧化应激因子的影响[J]. 河南医学研究, 2018, 27(12): 2201-2202. https://www.cnki.com.cn/Article/CJFDTOTAL-HNYX201812038.htm
[25] 胡艺丽, 李媛媛, 杨勤. 溃疡性结肠炎黏膜愈合相关生物标志物的研究进展[J]. 实用医学杂志, 2018, 34(21): 3644-3647. https://www.cnki.com.cn/Article/CJFDTOTAL-SYYZ201821040.htm
[26] Sakuraba A, Nemoto N, Hibi N, et al. Extent of disease affects the usefulness of fecal biomarkers in ulcerative colitis[J]. BMC Gastroenterol, 2021, 21(1): 197-204.
[27] Fukunaga S, Kuwaki K, Mitsuyama K, et al. Detection of calprotectin in inflammatory bowel disease: Fecal and serum levels and immunohistochemical localization[J]. Int J Mol Med, 2018, 41(1): 107-118.
[28] Chang JY, Cheon JH. Fecal immunochemical test and fecal calprotectin measurement are noninvasive monitoring tools for predicting endoscopic activity in patients with ulcerative colitis[J]. Gut Liver, 2018, 12(2): 117-118.
[29] Hiraoka S, Inokuchi T, Nakarai A, et al. Fecal immunochemical test and fecal calprotectin results show different profiles in disease monitoring for ulcerative colitis[J]. Gut Liver, 2018, 12(2): 142-148.
[30] Wakai M, Hayashi R, Tanaka S, et al. Serum amyloid A is a better predictive biomarker of mucosal healing than C-reactive protein in ulcerative colitis in clinical remission[J]. BMC Gastroenterol, 2020, 20(1): 85-93.
-




 下载:
下载: