Effects of Huoxue Tongjiang Prescription on intestinal flora and cysteine protease 3/gasdermin E pathway in rats with reflux esophagitis
-
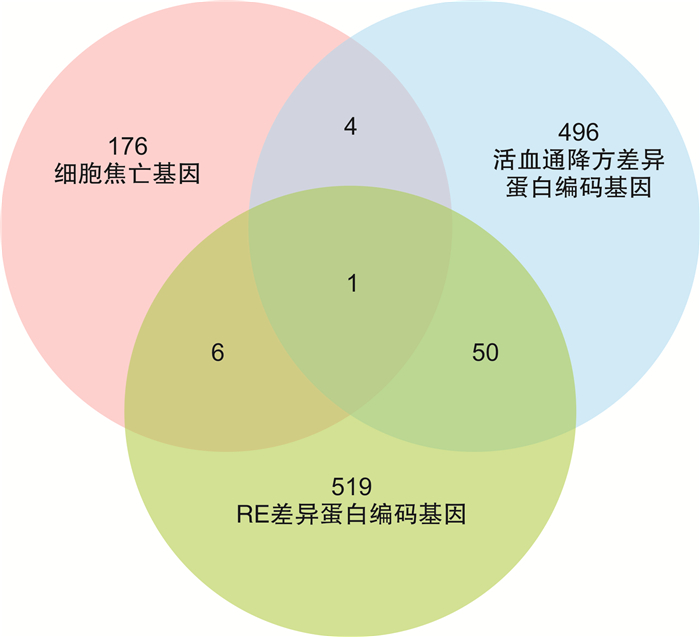
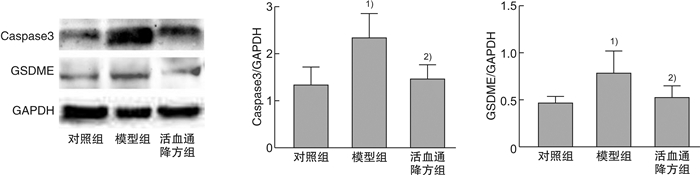
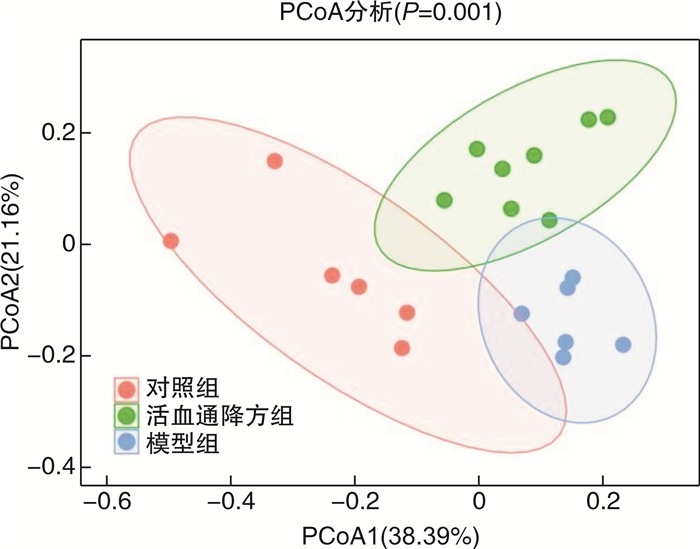
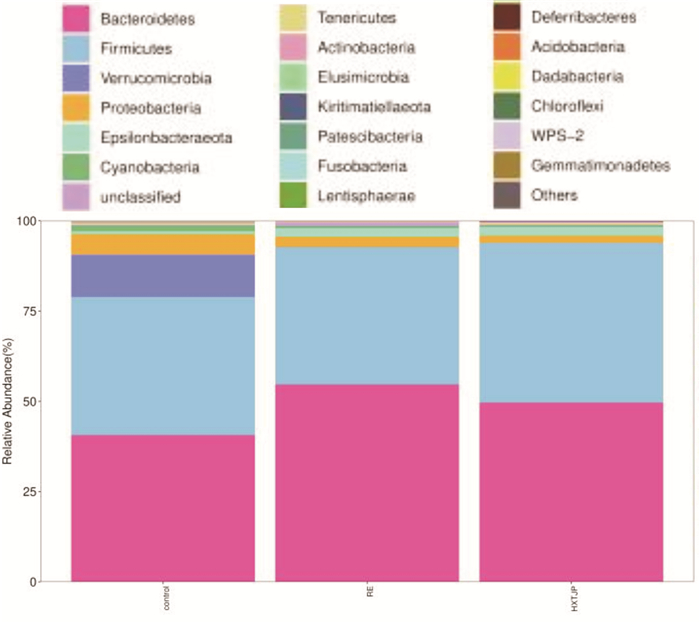
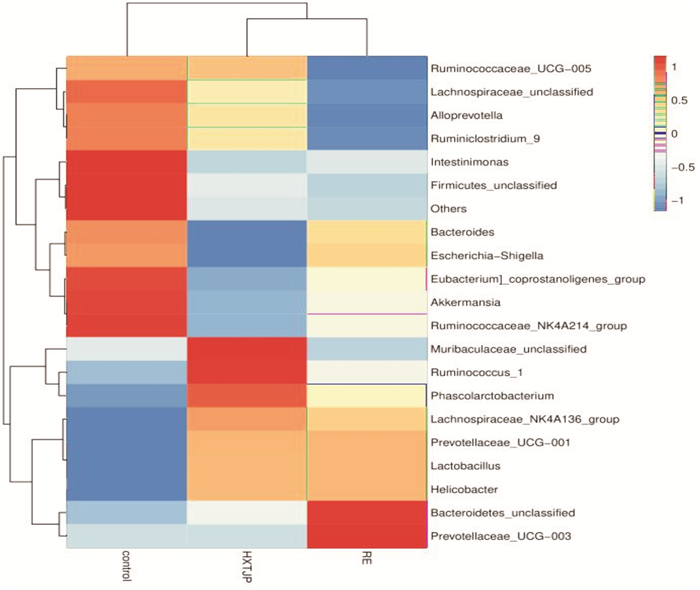
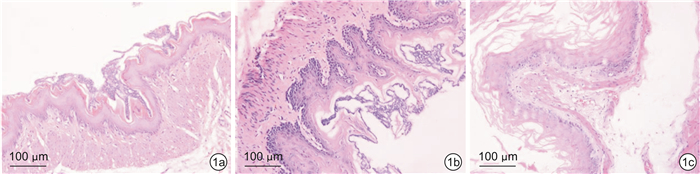
摘要: 目的 探讨活血通降方对反流性食管炎大鼠肠道菌群及半胱氨酸蛋白酶3/焦孔素E(Caspase-3/GSDME)通路的影响。方法 将30只Wistar大鼠随机分为对照组、模型组、活血通降方组,每组10只。除对照组外,其余两组均采用改良部分贲门肌切开术+外置幽门部分结扎术建立反流性食管炎大鼠模型。造模成功后,对照组及模型组予2.5 mL生理盐水灌胃,活血通降方组予2.5 mL活血通降方(3.68 g/kg)灌胃,2次/d,连续给药14 d。采用苏木精-伊红染色法观察各组大鼠食管组织病理变化;采用蛋白组学检测差异蛋白变化;采用酶联免疫吸附法检测血清中内毒素、IL-1β和IL-18的表达;采用蛋白免疫印迹法检测食管组织中Caspase-3和GSDME的表达,采用16S rDNA高通量测序检测肠道菌群的变化。结果 模型组大鼠食管组织病理评分较对照组增高(P<0.05),活血通降方组大鼠食管组织病理评分较模型组下降(P<0.05);蛋白组学分析表明,Caspase-3是各组的主要差异蛋白;与对照组比较,模型组肠道菌群的物种丰度显著降低,未分类拟杆菌属、Prevotellaceae_UCG-003属、螺旋菌属及Prevotellaceae_UCG-001属等革兰阴性菌的相对丰度显著增加,Akkermansia与Ruminococcaceae_UCG-005属的相对丰度显著降低,血清中内毒素、IL-1β和IL-18的含量及食管组织中Caspase-3和GSDME蛋白的表达明显升高(P<0.05,P<0.01);与模型组比较,活血通降方可改善肠道菌群组成,显著增加有益菌Ruminococcaceae_UCG-005的丰度,降低革兰阴性菌如Prevotellaceae_UCG-003属、拟杆菌属及未分类拟杆菌属的相对丰度,显著降低血清中内毒素、IL-1β和IL-18的含量,且可明显下调食管组织中Caspase-3和GSDME蛋白的表达水平(P<0.05,P<0.01)。结论 活血通降方可能通过调节肠道菌群降低体内内毒素水平,从而下调食管组织中Caspase-3和GSDME蛋白的表达及降低焦亡相关炎症因子的释放,对反流性食管炎大鼠黏膜损伤起保护作用。Abstract: Objective To investigate the effects of Huoxue Tongjiang Prescription on intestinal flora and cysteine protease 3/gasdermin E(Caspase-3/GSDME) pathway in rats with reflux esophagitis.Methods Thirty Wistar rats were randomly divided into control group(control group), model group(RE group) and Huoxue Tongjiang Prescription group, with 10 rats in each group. Except for the control group, all other groups used modified partial cardia myotomy and external pyloric partial ligation to establish RE rat models. After successful modeling, the control group and the Model group were given 2.5 mL normal saline intragastric administration, Huoxue Tongjiang Prescription group was given 2.5 mL Huoxue Tongjiang Prescription(3.68 g/kg) intragastric administration, twice a day for 14 days. The pathological changes of esophagus tissues were observed by hematoxylin-eosin(HE) staining. The changes of differential proteins were detected by proteomics. The expression of LPS, IL-1β, and IL-18 in serum was detected by Enzyme-linked immunosorbent assay(ELISA). The expression of Caspase-3 and GSDME in esophageal tissues was detected by protein immunoblotting. The changes of intestinal flora were detected by 16S rDNA high-throughput sequencing.Results Compared with the control group, the esophageal histopathological score of rats in the model group increased(P < 0.05); compared with the model group, the esophageal histopathological score of rats in the Huoxue Tongjiang Prescription group decreased(P < 0.05); proteomics analysis showed that Caspase-3 was the main differential protein in each group; compared with the control group, the species richness of the intestinal flora in the model group was significantly reduced, and the relative abundance of Gram-negative bacteria such as unclassified Bacteroides, Prevotellaceae_UCG-003, Helicobacter, and Prevotellaceae_UCG-001 were significantly increased. The relative abundance of Akkermansia and Ruminococcaceae_UCG-005 decreased significantly, the contents of LPS, IL-1β, and IL-18 in serum, and the protein expression of Caspase-3 and GSDME in esophageal tissues increased significantly(P < 0.05, P < 0.01). Compared with the model group, Huoxue Tongjiang Prescription can improve the composition of intestinal flora, significantly increase the abundance of beneficial bacteria Ruminococcaceae_UCG-005, and reduce the abundance of Gram-negative bacteria such as Prevotellaceae_UCG-003, Bacteroides and unclassified Bacteroides, and significantly reduce the content of LPS, IL-1β and IL-18 in serum, and significantly down-regulate the expression levels of Caspase-3 and GSDME protein in esophageal tissue(P < 0.05, P < 0.01).Conclusion Huoxue Tongjiang Prescription can reduce the level of LPS in the body by regulating the intestinal flora, thereby down-regulating the protein expression of Caspase-3 and GSDME in the esophageal tissue and reducing the release of pyroptosis-related inflammatory factors to protect the mucosal damage of rats with reflux esophagitis.
-

-
表 1 各组大鼠血清炎症因子LPS、IL-1β和IL-18含量的比较
X±S 组别 例数 剂量/(g·kg-1) LPS/(EU·L-1) IL-1β/(pg·mL-1) IL-18/(pg·mL-1) 对照组 5 0 265.45±20.36 748.46±367.05 254.14±22.90 模型组 5 0 363.16±24.831) 2 191.20±364.511) 420.94±73.691) 活血通降方组 5 3.68 288.84±9.322) 753.17±326.232) 289.11±35.632) 与对照组比较,1)P<0.01;与模型组比较,2)P<0.01。 表 2 各组大鼠肠道菌群α多样性指数分析
X±S 组别 例数 Observed otus chao1 shannon simpson 对照组 6 937.50±216.44 939.66±215.88 7.15±0.40 0.96±0.02 模型组 6 703.50±154.231) 704.81±155.451) 7.12±0.39 0.98±0.01 活血通降方组 8 815.50±189.28 817.58±191.00 7.56±0.49 0.99±0.01 与对照组比较,1)P<0.05。 表 3 各组大鼠肠道菌群属水平主要差异物种相对丰度比较
%,X±S 名称 对照组 模型组 活血通降方组 Prevotellaceae_UCG-003 0 5.15±3.392) 04) Akkermansia 11.71±10.33 0.06±0.072) 0.04±0.012) Bacteroides 5.86±3.40 3.01±4.60 0.30±0.172)3) Bacteroidetes_unclassified 0.28±0.26 12.40±5.262) 0.76±0.882)4) Lactobacillus 0.57±0.40 8.14±4.052) 7.89±5.302) Prevotellaceae_NK3B31_group 0.97±0.93 1.27±0.682) 1.63±1.272) Prevotellaceae_UCG-001 0.01±0.01 4.13±1.822) 4.07±2.002) Ruminococcaceae_UCG-005 2.42±1.50 0.21±0.132) 2.03±3.504) Eubacterium_coprostanoligenes_group 3.09±1.19 1.19±0.99 1.22±0.872) Helicobacter 0.71±0.82 2.31±1.471) 2.30±2.281) 与对照组比较,1)P<0.05,2)P<0.01;与模型组比较,3)P<0.05,4)P<0.01。 -
[1] 中华医学会消化病学分会. 2020年中国胃食管反流病专家共识[J]. 中华消化杂志, 2020, 40(10): 649-663. doi: 10.3760/cma.j.cn311367-20200918-00558
[2] Tack J, Pandolfino JE. Pathophysiology of Gastroesophageal Reflux Disease[J]. Gastroenterology, 2018, 154(2): 277-288. doi: 10.1053/j.gastro.2017.09.047
[3] D'Souza SM, Houston K, Keenan L, et al. Role of microbial dysbiosis in the pathogenesis of esophageal mucosal disease: A paradigm shift from acid to bacteria?[J]. World J Gastroenterol, 2021, 27(18): 2054-2072. doi: 10.3748/wjg.v27.i18.2054
[4] 王萍, 郑小领, 宋洁, 等. 胃食管反流患者肠道菌群、炎症变化及其与UCP2基因多态性的关系[J]. 临床和实验医学杂志, 2020, 19(19): 2049-2053. doi: 10.3969/j.issn.1671-4695.2020.19.009
[5] 张萌, 黄刚, 周志刚, 等. 反流对食管黏膜细胞凋亡相关基因表达的影响[J]. 河北医药, 2019, 41(17): 2580-2584. doi: 10.3969/j.issn.1002-7386.2019.17.004
[6] Wang Y, Gao W, Shi X, et al. Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a gasdermin[J]. Nature, 2017, 547(7661): 99-103. doi: 10.1038/nature22393
[7] Nadatani Y, Huo X, Zhang X, et al. NOD-Like Receptor Protein 3 Inflammasome Priming and Activation in Barrett's Epithelial Cells[J]. Cell Mol Gastroenterol Hepatol, 2016, 2(4): 439-453. doi: 10.1016/j.jcmgh.2016.03.006
[8] Yin XL, Wu HM, Zhang BH, et al. Tojapride prevents CaSR-mediated NLRP3 inflammasome activation in oesophageal epithelium irritated by acidic bile salts[J]. J Cell Mol Med, 2020, 24(2): 1208-1219. doi: 10.1111/jcmm.14631
[9] 柳媛, 刘菊, 刘亚婷, 等. 旋覆代赭汤对RE模型大鼠NLRP3/Caspase-1的影响[J]. 中国实验方剂学杂志, 2019, 25(20): 13-18. doi: 10.13422/j.cnki.syfjx.20191906
[10] 刘磊, 唐艳萍, 弓艳霞, 等. 活血通降方对反流性食管炎大鼠食管黏膜NF-κB/MAPK信号转导通路的影响[J]. 中国中西医结合外科杂志, 2021, 27(4): 555-562. doi: 10.3969/j.issn.1007-6948.2021.04.002
[11] 刘琰, 唐艳萍, 刘磊, 等. 活血通降方对反流性食管炎模型大鼠食管动力、血清炎症因子及食管下括约肌SCF/c-kit信号通路的影响[J]. 中医杂志, 2022, 63(3): 269-275. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZYZ202203013.htm
[12] 康丽丽, 唐艳萍, 杨莉, 等. 活血通降方对瘀血阻络型反流性食管炎临床疗效及胃排空的影响[J]. 中国中西医结合外科杂志, 2020, 26(4): 635-640. doi: 10.3969/j.issn.1007-6948.2020.04.011
[13] 陆星华, 张泰昌. 反流性食管炎诊断及治疗指南(2003年)[J]. 中华消化内镜杂志, 2004(4): 4-5. https://www.cnki.com.cn/Article/CJFDTOTAL-ZHXH200404000.htm
[14] 张声生, 朱生樑, 王宏伟, 等. 胃食管反流病中医诊疗专家共识意见(2017)[J]. 中国中西医结合消化杂志, 2017, 25(5): 321-326. http://zxpw.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=4f45188a-40fa-4b79-ac0a-4c2e0e91a783
[15] 胡颖超, 杨硕. 细胞焦亡的研究进展[J]. 南京医科大学学报(自然科学版), 2021, 41(8): 1245-1251. https://www.cnki.com.cn/Article/CJFDTOTAL-NJYK202108022.htm
[16] Liao XX, Dai YZ, Zhao YZ, et al. Gasdermin E: A Prospective Target for Therapy of Diseases[J]. Front Pharmacol, 2022, 13: 855828. doi: 10.3389/fphar.2022.855828
[17] Li L, Wang HH, Nie XT, et al. Sodium butyrate ameliorates lipopolysaccharide-induced cow mammary epithelial cells from oxidative stress damage and apoptosis[J]. J Cell Biochem, 2018.
[18] 陈梦, 戴海明. 细胞程序性死亡与炎症发生[J]. 中国细胞生物学学报, 2020, 42(12): 2205-2214. https://www.cnki.com.cn/Article/CJFDTOTAL-XBZZ202012013.htm
[19] 王玉鹏. 化疗药物激活caspase-3切割GSDME引发细胞焦亡[D]. 北京: 中国农业大学, 2019.
[20] Rogers C, Fernandes-Alnemri T, Mayes L, et al. Cleavage of DFNA5 by caspase-3 during apoptosis mediates progression to secondary necrotic/pyroptotic cell death[J]. Nat Commun, 2017, 8: 14128. doi: 10.1038/ncomms14128
[21] Barona I, Fagundes DS, Gonzalo S, et al. Role of TLR4 and MAPK in the local effect of LPS on intestinal contractility[J]. J Pharm Pharmacol, 2011, 63(5): 657-662. doi: 10.1111/j.2042-7158.2011.01253.x
[22] 张园, 彭伯坚, 骆凤娇. 菌群变化及脂多糖对胃食管反流病和Barrett食管的影响[J]. 广东医学, 2015, 36(11): 1773-1775. doi: 10.13820/j.cnki.gdyx.2015.11.045
[23] Tang YH, Liu HC, Song G, et al. A case-control study on the association of intestinal flora with ulcerative colitis[J]. AMB Express, 2021, 11(1): 106. doi: 10.1186/s13568-021-01267-9
[24] Deshpande NP, Riordan SM, Castano-Rodriguez N, et al. Signatures within the esophageal microbiome are associated with host genetics, age, and disease[J]. Microbiome, 2018, 6(1): 227. doi: 10.1186/s40168-018-0611-4
[25] Yang P, Feng W, Li C, et al. LPS induces fibroblast-like synoviocytes RSC-364 cells to pyroptosis through NF-kappa B mediated dual signalling pathway[J]. J Mol Histol, 2021, 52(4): 661-669. doi: 10.1007/s10735-021-09988-8
[26] Liu MJ, Yang JY, Yan ZH, et al. Recent findings in Akkermansia muciniphila-regulated metabolism and its role in intestinal diseases[J]. Clin Nutr, 2022, 41(10): 2333-2344. doi: 10.1016/j.clnu.2022.08.029
[27] Tuikhar N, Keisam S, Labala RK, et al. Comparative analysis of the gut microbiota in centenarians and young adults shows a common signature across genotypically non-related populations[J]. Mech Ageing Dev, 2019, 179: 23-35. doi: 10.1016/j.mad.2019.02.001
-




 下载:
下载: